New Product: Vivo Morpholino end modifications available now. Read about them here.
Citations of papers using Vivo-Morpholinos are here.
Why use a Vivo-Morpholino?
Vivo-Morpholinos* are the knockdown, exon-skipping or miRNA blocking reagent of choice for in vivo experiments. Outstanding results can be achieved systemically with intravenous (I.V.) injection, and modest systemic delivery achieved with Vivo-Morpholinos by intraperitoneal (I.P.) injection. Efficient localized delivery can be achieved by injecting the Vivo-Morpholino directly into the area of interest. Vivo-Morpholinos are also effective and easy to use in cultures.
Vivo-Morpholinos get the job done by combining two important parts
A Vivo-Morpholino is comprised of a Morpholino oligo with a unique covalently linked delivery moiety, which is comprised of an octa-guanidine dendrimer. It uses the active component of arginine rich delivery peptides (the guanidinium group) with improved stability and reduced cost. The Vivo-Morpholino is assembled by coupling the vivo-delivery group to a Morpholino while the oligo is still bound to its synthesis resin, allowing excellent purification by washing the solid-phase resin. Vivo-Morpholinos must be chosen prior to synthesis and cannot be added later because the vivo-delivery group is added to a Morpholino prior to cleavage from its synthesis resin.
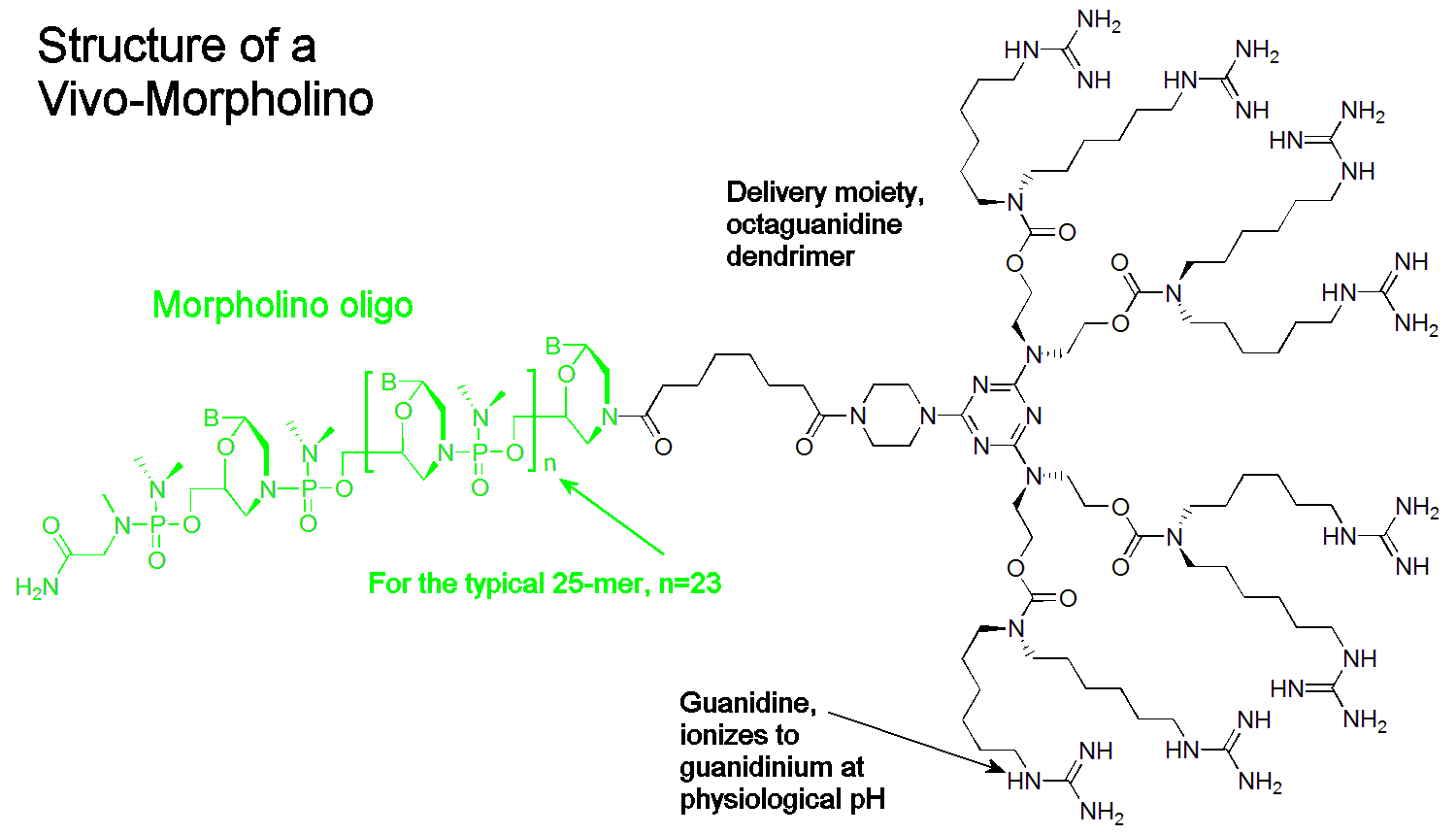 Vivo-Morpholinos are offered in three convenient amounts
Vivo-Morpholinos are offered in three convenient amountsAll Vivo-Morpholinos are provided in 400, 2000, and 10000 nmole quantities in lyophilized form. We suggest you make a 0.5 mM solution in phosphate buffered saline; while solubility varies and is sequence-dependent, higher concentrations risk exceeding the solubility of the Vivo-Morpholino. For syntheses greater than 10000 nmol please contact Customer Support. All Vivo-Morpholinos are sterilized. The amount of oligo you will need depends on the experiment and the method of injection. For a typical 20g mouse injected at 12.5 mg/kg I.V. the dose is approximately 25 nmole per injection. This means you will get approximately 16 injections from the 400 nanomole quantity, 80 injections from the 2000 nanomole quantity and 400 injections from the 10000 nanomole quantity.
In vivo methods
For best systemic delivery results the injection method of choice is i.v. although i.p. can also be used. The maximum suggested dosage in mammals is 12.5 mg/kg in a 24 hour period; dosing can be repeated daily, however, the sensitivity of organisms to the toxicity of the delivery moiety can vary depending on the type and health of the organism, so it is important to check the tolerance of your organism prior to dosing a group of animals. Local injections should be performed with reduced doses.
For short term experiments (3-day) Gene Tools suggests two days of i.v. injections at 12.5 mg/kg followed by analysis on day 3. Long term experiments should be optimized for your specific goals; however you can try starting with a loading dose equivalent to the short term experiment and then adjust dosage and times to find an optimum for continued delivery. Vivo-Morpholinos were optimized using 4 to 10 week old C57 Black mice and up to 20 mg/kg injections with successful outcomes. However, we have found that younger or older mice do not tolerate Vivo-Morpholinos as well and may require substantially lower doses. In addition the use of compromised mice, such as those with less robust genetic backgrounds, may require further dose limitations.
Cell culture methods
Vivo-Morpholinos are very simple to use in cultured cells. The typical concentration range for Vivo-Morpholinos in cell culture is 1 to 10 micromolar of Morpholino oligo in the culture medium. Concentrations higher than 10 micromolar could be toxic to cells. The protocol is as follows: add an appropriate amount of the solution to the culture medium on cells to reach the desired final concentration of Morpholino oligo, swirl to mix and harvest the cells as early as 24hr after treatment. It is not necessary to change the culture medium after adding oligos if the oligos are not toxic to the cells. If toxicity is observed, the oligo containing medium can be removed 2 to 4 hrs after treatment and fresh medium added. Serum significantly inhibits the efficacy of Vivo-Morpholinos so use a lower serum concentration if the cells tolerate it. The effect of splice-targeted oligos can be detected by reverse transcriptase PCR. The effect of translation blocking oligos can be assessed by analyzing protein levels by Western blot. The time needed to detect changes in the amount of protein translated from the targeted RNA will vary depending on the half-life of the protein, as preexisting protein must degrade for a knockdown to be evident.
Delivery to most major organs is achieved
Following the short-term delivery experiment described above, one can expect quantifiable knockdown or exon-skipping in liver, small intestine, colon, muscle, lung, and stomach tissues with lesser but quantifiable delivery in the spleen, heart, skin and brain. For assessment of specific tissues within an organ one can achieve greater results with direct localized injections at the same concentrations described. Brain delivery is far more effective by introducing Vivo-Morpholinos directly into the cerebrospinal fluid, as by intracerebroventricular infusion (i.c.v.) or direct injection into a specific brain region.
Vivo-Morpholinos are stable
Vivo-Morpholinos are provided sterile and are best stored at room temperature. If your Vivo-Morpholino becomes contaminated, it can be filter sterilized or autoclaved. Some of the Vivo-Morpholino will degrade on autoclaving (loss of delivery dendrimer); we recommend only autoclaving a given Vivo-Morpholino once to limit loss of potency. To autoclave, disable the autoclave's vacuum cycle to prevent loss of liquid. To filter sterilize, use a 0.2 micron polysulfone membrane; do not use other membranes as they have higher affinity for Vivo-Morpholinos and their use can result in significant loss of oligo concentration. To minimize decrease in oligo concentration due to association with the membrane, use the smallest available syringe filter (13 mm or smaller).
NEW! Vivo-Morpholino 5' Modification
The in vivo delivery moiety is an octoguanidine dendrimer which conjugates to the 3' end of a Morpholino oligo to enhance cell penetration. Gene Tools offers further modification to the 5' end of Vivo-Morpholinos. Vivo Morpholinos with these end modifications are sold in 400nmol, 2000nmol, and 10,000nmol quantities
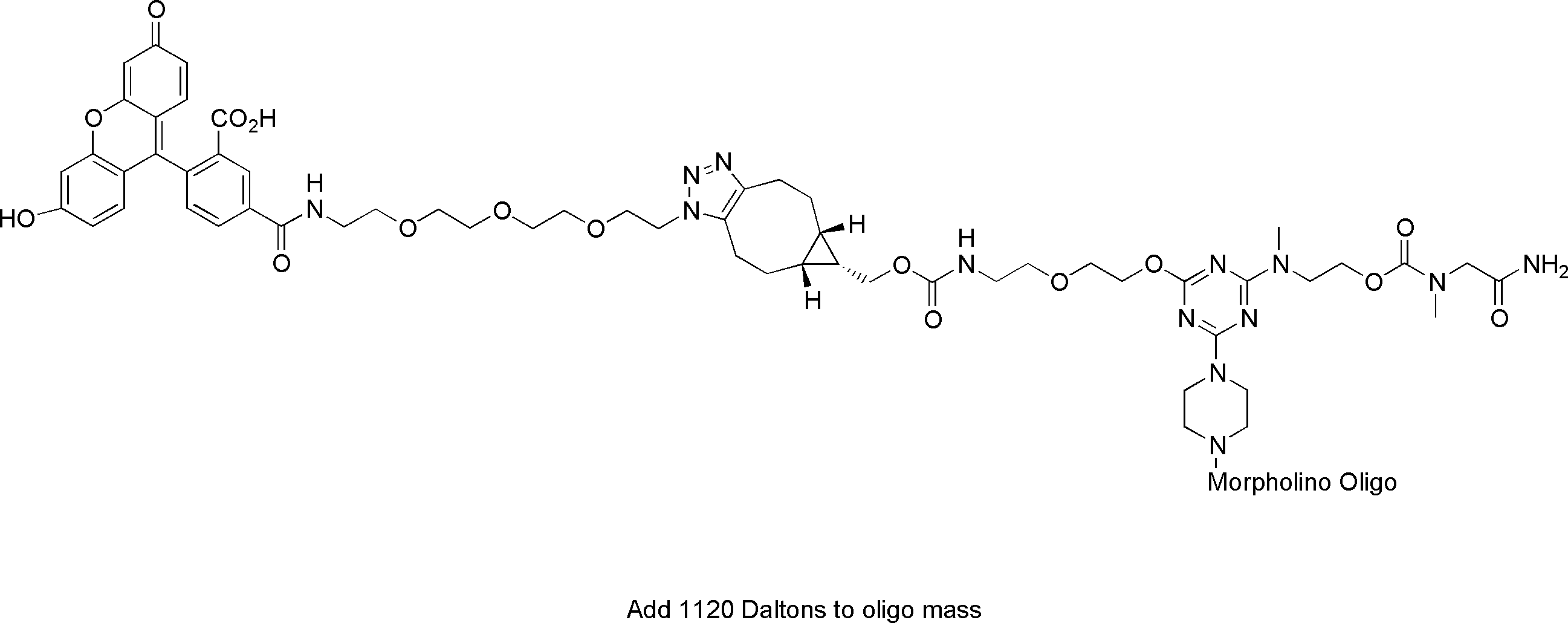
Fluorescein Labeled Vivo-Morpholino
The Fluorescein labeled Vivo-Morpholino is assembled using click chemistry to covalently attach a water soluble carboxy fluorescein to the 5' end of the Vivo Morpholino. Cell delivery of the Fluorescent Vivo-Morpholino can be visualized by fluorescence microscopy.
Excitation Peak: 501.5nm
Emission Peak: 525.5nm
Adds 1120 daltons to the molecular mass of the Vivo-Morpholino.
Code: 5F-V
Structure:
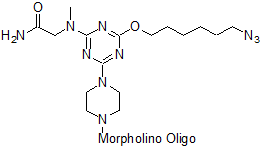
Azide Modified Vivo-Morpholino:
Azide-Vivo-Morpholinos contain an azide functional group to the 5' end of the Vivo-Morpholino. The Azide-Vivo-Morpholino may have further modifications added off column using click chemistry. Further 5' end modification of the Vivo-Morpholino should consider the hydrophilicity and total positive charges of the resultant conjugate as the former may affect its aqueous solubility and the latter may impact its in-vivo delivery efficiency.
Adds 304 daltons to the oligo mass of the Vivo-Morpholino.
Code: 5Z-V
Structure:
Vivo-Morpholinos can be ordered online
You can log into our online store and order Vivo-Morpholinos (to log in, see the box in the left column on this page). For more information on how to order Vivo-Morpholinos Contact Us.
*US Patent 7,935,816
References
For an open-access introduction to Vivo-Morpholinos, see:
Paul A. Morcos, Yongfu Li, and Shan Jiang. Vivo-Morpholinos: A non-peptide transporter delivers Morpholinos into a wide array of mouse tissues. BioTechniques. 2008 Dec;45(6):616-26.
Balashova OA, Panoutsopoulos AA, Visina O, Selhub J, Knoepfler PS, Borodinsky LN. Noncanonical function of folate through folate receptor 1 during neural tube formation. Nat Comm. 2024;15:1642. doi:10.1038/s41467-024-45775-1
cell culture: WTC-11 hiPSC
Dou Z, Lei H, Su W, Zhang T, Chen X, Yu B, Zhen X, Si J, Sun C, Zhang H, Di C. Modification of BCLX pre-mRNA splicing has antitumor efficacy alone or in combination with radiotherapy in human glioblastoma cells. Cell Death Dis. 2024;15:160. doi:10.1038/s41419-024-06507-x
cell culture: A172, normal astrocyte cells (HA1800)
Patel JH, Angell Swearer A, Kakebeen AD, Loh LR, Wills AE. Protocol for tail vein injection in Xenopus tropicalis tadpoles. STAR Protoc. 2024 Feb 16;5(1):102895. doi: 10.1016/j.xpro.2024.102895. Online ahead of print.
Pucci G, Minafra L, Bravatà V, Calvaruso M, Turturici G, Cammarata FP, Savoca G, Abbate B, Russo G, Cavalieri V, Forte GI. Glut-3 Gene Knockdown as a Potential Strategy to Overcome Glioblastoma Radioresistance. Int J Mol Sci. 2024;25(4):2079; doi:10.3390/ijms25042079
Noor SM, Wong CED, Wong PF, Norazit A. Generation of glial cell-derived neurotrophic factor (gdnf) morphants in zebrafish larvae by cerebroventricular microinjection of vivo morpholino. Methods Cell Biol. 2024;181:17-32. doi: 10.1016/bs.mcb.2022.09.004. Epub 2022 Oct 31. "The morpholino knockdown was performed in situ in 4 days post fertilization (dpf) larvae via cerebroventricular microinjection of gdnf vivo morpholino."
Damacena de Angelis C, Meddeb M, Chen N, Fisher SA. An anti-sense oligonucleotide efficiently suppresses splicing of an alternative exon in vascular smooth muscle in vivo. Am J Physiol Heart Circ Physiol. 2024 Jan 26. doi: 10.1152/ajpheart.00745.2023. Online ahead of print.
"In contrast an octo-guanidine conjugated anti-sense oligonucleotide targeting the 5' splice site of Mypt1 E24 was highly efficient in vivo. It reduced the percent splicing inclusion of Mypt1 E24 from 80 to 10% in mesenteric arteries. The maximal and half-maximal effects occurred at 12.5 and 6.25 mg/kg, respectively. The effect persisted for at least one month without toxicity. This highly effective splice blocking anti-sense oligonucleotide could be developed as a novel therapy to reverse vascular dysfunction common to diseases such as hypertension and heart failure."
"Prior to injection the ASOs were diluted in saline to give the appropriate dose and 133 injected intraperitoneally. Control mice received either the control ASO or vehicle. For dose-134 response assays adult male and female mice received three doses every other day of ASO at 135 0.2, 1.0, 6.25 or 12.5 mg/kg, and euthanized the next day. For time course analysis male and 136 female mice were treated with 12.5mg/Kg ASO every other day for three doses and euthanized 137 on days 1, 4, 7, 14 or 28 after the last dose."
Gebru NT, Hill SE, Blair LJ. Development of FKBP5 ASOs to mitigate tau pathology. Alzheimers Dement. 2023;[Epub] doi:10.1002/alz.082780
Shihabeddin E, Santhanam A, O'Brien J. Cost-effective strategies to knock down genes of interest in the retinas of adult Zebrafish. Front Cell Neurosci. 2023;(17):[Epub] doi:10.3389/fncel.2023.1321337
Chatterjee R, Shukla A, Chakrabarti K, Chatterji U. CLEC12A sensitizes differentially responsive breast cancer cells to the anti-cancer effects of artemisinin by repressing autophagy and inflammation. Front Oncol. 2023 Dec 8;13:1242432. doi: 10.3389/fonc.2023.1242432. eCollection 2023.
"Murine mammary tumors were allowed to develop in BALB/c mice injected with 10^7 4T1 cells. Tumor-bearing mice were injected intravenously with vivo-morpholino for TLR4 (VMO-TLR4; 5’ CAGGGAGGCATCATCCTGGCATTTT 3’) or scramble morpholino (SC-MO) every other day for a week (n=3), along with ART (25 mg/kg) for 7 consecutive days".
Shiba N, Yang X, Sato M, Kadota S, Suzuki Y, Agata M, Nagamine K, Izumi M, Honda Y, Koganehira T, Kobayashi H, Ichimura H, Chuma S, Nakai J, Tohyama S, Fukuda K, Miyazaki D, Nakamura A, Shiba Y. Efficacy of exon-skipping therapy for DMD cardiomyopathy with mutations in actin binding domain 1. Mol Ther Nucleic Acids. 2023 Oct 19;34:102060. doi: 10.1016/j.omtn.2023.102060. eCollection 2023 Dec 12.
"On day 36, hiPSC-CMs were treated with Vivo-Morpholinos (0.5, 1.0, 2.0, and 4 μM in RPMI/B27+ins) and S.C. (4 μM in RPMI/B27+ins) for 12 h. The cells were maintained in RPMI/B27+ medium and changed every 3 d for 14 d. On day 60, hiPSC-CMs were reseeded on glass-bottom dishes for imaging analysis, fixed with 4% paraformaldehyde for immunofluorescence staining, lysed with RNeasy Mini Kit for mRNA analysis, and lysed with the M-PER Mammalian Protein Extraction Reagent for western blotting analysis".
Xu L, Zhao M, Zohar Y, Wong T-T. Induction of Reproductive Sterility in Coho Salmon (Oncorhynchus kisutch) by an Immersion-Based Gene Silencing Technology. J Mar Sci Eng. 2023;11:2208. doi:10.3390/jmse11122208
"After washing with the wash medium a few times, eggs were immersion-treated in different immersion medium containing 12.5 to 20 µM csdnd-MO-Vivo for 12 to 36 h at 4 °C or 8 °C with gentle shaking (at around 70 rpm depending on immersion volume) over an orbital shaker. About 200 to 400 eggs were treated for each group in sealed 2-cup glass bowls with 25–50 mL immersion medium."
Liu C, Liu X, He Z, Zhang J, Tan X, Yang W, Zhang Y, Yu T, Liao S, Dai L, Xu Z, Li F, Huang Y, Zhao J. Proenkephalin-A secreted by renal proximal tubules functions as a brake in kidney regeneration. Nat Commun. 2023 Nov 7;14(1):7167. doi: 10.1038/s41467-023-42929-5.
"..tcf21 vivo-MO (25 μM, 10 μL per fish) or penka vivo-MO (100 μM, 10 μL per fish) was intraperitoneally injected into zebrafish at 2, 4, and 6 dpi, and kidneys were collected at 7 dpi for RNA extraction and RT-PCR..."
Al Qaryoute A, Fallatah W, Dhinoja S, Raman R, Jagadeeswaran P. Role of microRNAs and their downstream target transcription factors in zebrafish thrombopoiesis. Sci Rep. 2023 Sep 26;13(1):16066. doi: 10.1038/s41598-023-42868-7.
DNA antisense was annealed (piggybacked) on Vivo-Morpholino for delivery and injected into adult zebrafish.
Sundaramoorthi H, Fallatah W, Mary J, Jagadeeswaran P. Discovery of seven hox genes in zebrafish thrombopoiesis. Blood Cells Mol Dis. 2023 Aug 30;104:102796. doi: 10.1016/j.bcmd.2023.102796. Online ahead of print.
DNA antisense was annealed (piggybacked) on Vivo-Morpholino for delivery and injected into adult zebrafish.
Ang Z, Paruzzo L, Hayer KE, Schmidt C, Torres-Diz M, Xu F, Zankharia U, Zhang Y, Soldan SS, Zheng S, Falkenstein CD, Loftus JP, Yang SY, Asnani M, King Sainos P, Pillai V, Chong ER, Li M, Tasian SK, Barash Y, Lieberman PM, Ruella M, Schuster SJ, Thomas-Tikhonenko A. Alternative splicing of its 5' UTR limits CD20 mRNA translation and enables resistance to CD20-directed immunotherapies. Blood. 2023 Sep 8:blood.2023020400. doi: 10.1182/blood.2023020400. Online ahead of print.
cell culture: OCI-Ly8, Raji, and MEC-1
Fang J, Wang J, Wang Y, Liu X, Chen B, Zou W. Ribo-On and Ribo-Off tools using a self-cleaving ribozyme allow manipulation of endogenous gene expression in C. elegans. Commun Biol. 2023;6:816. doi:10.1038/s42003-023-05184-4
"We injected v-M8 into the argn-1::gfp::Rz animals on day 1 of adulthood and detected the protein expression after 48 h (Fig. 5d). Quantitative analysis of ARGN-1::GFP levels revealed that endogenous argn-1 expression was partially recovered in a dose-dependent manner. Injection of morpholino at 250 μM successfully restored the gene expression to levels comparable to those of the inactive T3H38 control group, suggesting an efficient block of the ribozyme-mediated self-cleavage"
Dery KJ, Kojima H, Kageyama S, Kadono K, Hirao H, Cheng B, Zhai Y, Farmer DG, Kaldas FM, Yuan X, Eltzschig HK, Kupiec-Weglinski JW. Alternative splicing of CEACAM1 by hypoxia-inducible factor-1α enhances tolerance to hepatic ischemia in mice and humans. Sci Transl Med. 2023 Aug 2;15(707):eadf2059. doi: 10.1126/scitranslmed.adf2059. Epub 2023 Aug 2. PMID: 37531413
Arumi-Planas M, Rodriguez-Baena FJ, Cabello-Torres F, Gracia F, Lopez-Blau C, Nieto MA, Sanchez-Laorden B. Microenvironmental Snail1-induced immunosuppression promotes melanoma growth. Oncogene. 2023 Jul 29. doi: 10.1038/s41388-023-02793-5. Online ahead of print.
"C57BL/6 mice aged 7 weeks were injected in the tail vein with BrafV600E-5555 -Luciferase cells. 10 dpi, a solution containing Snail1-MO or Control-MO in saline (100 μl; 6 mg MO per kg) was injected in the tail vein of the corresponding mice every other day. After 20 days mice were sacrificed and lungs were processed, sectioned, and subjected to analysis."
Balashova OA, Panoutsopoulos AA, Visina O, Selhub J, Knoepfler PS, Borodinsky LN. Non-canonical function of folate/folate receptor 1 during neural tube formation. bioRxiv. 2023 Jul 21:[preprint]. doi:10.1101/2023.07.19.549718.
"hiPSC-derived embryoid bodies were incubated with 2 μM control-vivo-morpholino (Control), FOLR1-vivo morpholino (FOLR1476 knockdown (KD), FOLR1-MO) or FOLR1-MO and 50 μM pteroate (FOLR1 KD + pteroate) until 18-21 days in vitro."
He S, Song W, Cui S, Li J, Jiang Y, Chen X, Peng L. Modulation of miR-146b by N6-methyladenosine modification remodels tumor-associated macrophages and enhances anti-PD-1 therapy in colorectal cancer. Cell Oncol (Dordr). 2023 Jul 5. doi: 10.1007/s13402-023-00839-0. Online ahead of print.
"In some experiments, morpholino (MO) was dissolved in PBS, and WT BMDMs were preincubated with MO–nc (5 μM) and MO-methyltransferase-like 3 (Mettl3) (5 μM) for 12 h, polarized to the M2 phenotype, and then mixed with MC38 tumor cells. All adoptive transfer experiments were carried out in new WT mice."
Qaryoute AA, Fallatah W, Dhinoja S, Raman R, Jagadeeswaran P. Role of MicroRNAs and their Downstream Target Transcription Factors in Zebrafish Thrombopoiesis. Res Sq. 2023 Apr 24:rs.3.rs-2807790. doi: 10.21203/rs.3.rs-2807790/v1. Preprint.
Delivery of DNA oligos by Vivo-Morpholino piggyback
Haas M, Caron G, Chatonnet F, Manenti S, Alaterre E, Devin J, Delaloy C, Bertolin G, Viel R, Pignarre A, Llamas-Gutierrez F, Marchalot A, Decaux O, Tarte K, Delpy L, Moreaux J, Fest T. PIM2 kinase has a pivotal role in plasmablast generation and plasma cell survival, opening up novel treatment options in myeloma. Blood. 2022;139(15):2316-37. doi:10.1182/blood.2021014011
Delivery in culture to Plasmoblasts (PBs) and CD23+ post-activated B cells
Sheng P, Li L, Li T, Wang Y, Hiers NM, Mejia JS, Sanchez JS, Zhou L, Xie M. Screening of Drosophila microRNA-degradation sequences reveals Argonaute1 mRNA's role in regulating miR-999. Nat Commun. 2023 Apr 13;14(1):2108. doi: 10.1038/s41467-023-37819-9.
"6 × 106 S2 cells/well were seeded in six-well plates and incubated with 1 µM, 2.5 µM or 5 µM vivo-morpholino oligos against AGO1 TDMD trigger in the medium for 48 h, then the total RNA was extracted for northern blot and RT-qPCR."
Wan Y, Slevin E, Koyama S, Huang CK, Shetty AK, Li X, Harrison K, Li T, Zhou B, Lorenzo SR, Zhang Y, Salinas JM, Xu W, Klaunig JE, Wu C, Tsukamoto H, Meng F. miR-34a regulates macrophage-associated inflammation and angiogenesis in alcohol-induced liver injury. Hepatol Commun. 2023 Apr 4;7(4):e0089. doi: 10.1097/HC9.0000000000000089. eCollection 2023 Apr
miRNA target in mice
Taghi Khani A, Kumar A, Sanchez Ortiz A, Radecki C, Aramburo S, Lee SJ, Hu Z, Damirchi B,Lorenson MY, Wu X, Gu Z, Stohl W, Sanz I, Meffre E, Müschen M, Forman SJ, Koff JL, Walker AM. Swaminathan S. Isoform-specific knockdown of long and intermediate prolactin receptors interferes with evolution of B-cell neoplasms. Commun Biol. 2023;6:295. doi:10.1038/s42003-023-04667-8
NOD-SCID IL2Rγ−/− (NSG) mice, xenografted, implanted with Alzet osmotic minipumps to treat with control or splice-modulating Vivo-Morpholino for 8 weeks
Kundu D, Kennedy L, Zhou T, Ekser B, Meadows V, Sybenga A, Kyritsi K, Chen L, Ceci L, Wu N, Wu C, Glaser S, Carpino G, Onori P, Gaudio E, Alpini G, Francis H. p16 INK4A drives non-alcoholic fatty liver disease phenotypes in high fat diet fed mice via biliary e2f1/foxo1/igf-1 signaling. Hepatology. 2023 Feb 20. doi: 10.1097/HEP.0000000000000307. Online ahead of print.
"4 wk wild-type (WT, C57BL/6J) male mice were fed control diet (CD) or HFD and received either p16 or control Vivo Morpholino (VM) by tail vein injection 2x during the 16th wk of feeding."
Sugitani K, Mokuya T, Homma S, Maeda M, Konno A, Ogai K. Specific Activation of Yamanaka Factors via HSF1 Signaling in the Early Stage of Zebrafish Optic Nerve Regeneration. Int J Mol Sci. 2023;24(4):3253. doi:10.3390/ijms24043253
"The MOs were injected into the eye with a Hamilton 33G neuron syringe. Twenty hours after the injection of 0.75 µL of MO solution (0.5 mM) into the eye, the optic nerve was crushed."
Cheng Y, Zhang Z, Zhang G, Chen L, Zeng C, Liu X, Feng Y. The Male-Biased Expression of miR-2954 Is Involved in the Male Pathway of Chicken Sex Differentiation. Cells. 2022 Dec 20;12(1):4. doi: 10.3390/cells12010004.
"The morpholino was resuspended in sterile phosphate-buffered saline (PBS) to form a 0.5-mM stock solution. Before microinjection, VMO-miR-2954 was prepared into 1 μg/μL with PBS. For the best systemic delivery results from blood vessels, the fertilized eggs were pre-incubated to embryonic day 3.0 and placed on a 2-cm square adhesive tape near the equator. A small hole was cut in the eggshell and 3 μL VMO-miR-2954 solution was injected into the major blood vessel using a Sutter-pulled glass capillary needle with the hand-held microsyringe under the stereomicroscope (MZ75, Leica, German). Eggs were sealed with clear tape and incubation was resumed. Blood was collected from yolk sac vessels at E6.0 for sex determination and then continued to hatch to desired stages."
Jonusaite S, Oulhen N, Izumi Y, Furuse M, Yamamoto T, Sakamoto N, Wessel G, Heyland A. Identification of the genes encoding candidate septate junction components expressed during early development of the sea urchin, Strongylocentrotus purpuratus, and evidence of a role for Mesh in the formation of the gut barrier. Dev Biol. 2022 Dec 29;495:21-34. doi: 10.1016/j.ydbio.2022.12.007. Online ahead of print.
"For initial vMO treatments,late gastrula stage embryos (~48hpf, when Mesh mRNA transcripts are first detected by scRNA seq...) were collected in 1.5 ml microtubes and exposed to 500 μl FASW containing 1 μM, 2.5 μM and 5 μM Spmesh or control vMO, or milliQ water (as a carrier control used to dilute vMO stock solutions). The embryos were incubated for 24h at 14 0C to allow them to reach the early pluteus stage (72hpf) as this is the oldest stage used in all of the experiments. We chose 2.5 μM vMO concentration as the final working concentration since exposure to5 μM Spmesh or control vMO reduced larval swimming activity (the larvae looked normal otherwise;data not shown),suggesting a non-specific effect of this concentration of vMOs on early larvae."
Arumi-Planas M, Rodriguez-Baena FJ, Cabello-Torres F, Gracia F, Lopez-Blau C, Nieto MA, Sanchez-Laorden B. Microenvironmental Snail1 is a driver of immunosuppression in melanoma. bioRxiv. 2022;[preprint] doi:10.1101/2022.11.23.517623
"C57BL/6 mice aged 7 weeks were injected in the tail vein with BrafV600E-5555 -Luciferase cells. 10 dpi, a solution containing Snail1-MO or Control-MO in saline (100 μl; 6 mg MO per kg) was injected in the tail vein of the corresponding mice every other day. After 20 days mice were sacrificed and lungs were processed, sectioned, and subjected to analysis."
Meadows V, Marakovits C, Ekser B, Kundu D, Zhou T, Kyritsi K, Pham L, Chen L, Kennedy L, Ceci L, Wu N, Carpino G, Zhang W, Isidan A, Meyer A, Owen T, Gaudio E, Onori P, Alpini GD, Francis H. Loss of Apical Sodium Bile Acid Transporter Disrupts Bile Acid Circulation and Reduces Biliary Damage in Cholangitis. Am J Physiol Gastrointest Liver Physiol. 2022 Nov 21. doi: 10.1152/ajpgi.00112.2022. Online ahead of print.
mice
Zhu P, Zheng P, Kong X, Wang S, Cao M, Zhao C. Rassf7a promotes spinal cord regeneration and controls spindle orientation in neural progenitor cells. EMBO Rep. 2022 Nov 21:e54984. doi: 10.15252/embr.202254984. Online ahead of print.
Liang H, Li Y, Li M, Zhou W, Chen J, Zhang Z, Yang Y, Ran C, Zhou Z. The effect and underlying mechanism of yeast β-glucan on antiviral resistance of zebrafish against spring viremia of carp virus infection. Front Immunol. 2022 Nov 3;13:1031962. doi: 10.3389/fimmu.2022.1031962. eCollection 2022.
"Zebrafish larvae (4dpf) were added with 25 nmol/L of CRFB1 and CRFB2 vivo MO (CRFB1 and CRFB2 vivo MO were added together during the experiment), or standard control vivo MO, and then treated ..."
Zhu X, Liu J, Cai M, Bao L, Pan Y, Wu P, Chu W, Zhang J. The circadian rhythm regulates branched-chain amino acids metabolism in fast muscle of Chinese perch (Siniperca chuatsi) during short-term fasting by Clock-KLF15-Bcat2 pathway. Br J Nutr. 2022 Nov 14:1-31. doi: 10.1017/S0007114522003646. Online ahead of print.
Zhang Y, Zhang L-S, Dai Q, Chen P, Lu M, Kairis EL, Murugaiah V, Xu J, Shukla RK, Liang X, Zou Z, Cormet-Boyaka E, Qiu J, Peeples ME, Sharma A, He C, Li J. 5-methylcytosine (m5C) RNA modification controls the innate immune response to virus infection by regulating type I interferons. Proc Nat Acad Sci USA. 2022;119(42):e2123338119 doi:10.1073/pnas.2123338119
Delivery into mice with Vivo-Morpholinos by intranasal & i.v. injection routes
Joy E. Neuromuscular NMDA receptors regulate developmental synaptic depletion. Mult Scler. 2022;9(5):442 doi:10.35248/2376-0389.22.9.5.442
Al-Dabet MM, Shahzad K, Elwakiel A, Sulaj A, Kopf S, Bock F, Gadi I, Zimmermann S, Rana R, Krishnan S, Gupta D, Manoharan J, Fatima S, Nazir S, Schwab C, Baber R, Scholz M, Geffers R, Mertens PR, Nawroth PP, Griffin JH, Keller M, Dockendorff C, Kohli S, Isermann B. Reversal of the renal hyperglycemic memory in diabetic kidney disease by targeting sustained tubular p21 expression. Nat Commun. 2022;13,5062. doi:10.1038/s41467-022-32477-9
"DNMT1 vivo morpholino (DNMTi, targeting the translation of transcript variant 1 of mouse DNMT1; 6 mg/kg body weight in PBS, every other day, intraperitoneally)"
Magri F, Zanotti S, Salani S, Fortunato F, Ciscato P, Gerevini S, Maggi L, Sciacco M, Moggio M, Corti S, Bresolin N, Comi GP, Ronchi D. Antisense Morpholino-Based In Vitro Correction of a Pseudoexon-Generating Variant in the SGCB Gene. Int J Mol Sci. 2022;23(17):9817. doi:10.3390/ijms23179817
Kruyer A, Angelis A, Garcia-Keller C, Li H, Kalivas PW. Plasticity in astrocyte subpopulations regulates heroin relapse. Sci Adv. 2022 Aug 12;8(32):eabo7044. doi: 10.1126/sciadv.abo7044. Epub 2022 Aug 10.
"...animals were fitted with bilateral cannulae above the NAcore (+1.5 mm AP, ±1.8 mm ML, and −5.5 mm DV). Starting on day 6 of extinction training, animals received infusions ... 1.5 mm beyond the base of the guide cannulae (50 μM, 1 μl per hemisphere, and 0.5 μl/min) for three consecutive days."
Aljiboury A, Mujcic A, Curtis E, Cammerino T, Magny D, Lan Y, Bates M, Freshour J, Ahmed-Braimeh YH, Hehnly H. Pericentriolar matrix (PCM) integrity relies on cenexin and polo-like kinase (PLK)1. Mol Biol Cell. 2022 Aug 1;33(9):br14. doi: 10.1091/mbc.E22-01-0015. Epub 2022 May 24.
Cripe TP, Hutzen B, Currier MA, Chen CY, Glaspell AM, Sullivan GC, Hurley JM, Deighen MR, Venkataramany AS, Mo X, Stanek JR, Miller AR, Wijeratne S, Magrini V, Mardis ER, Mendell JR, Chandler DS, Wang PY. Leveraging gene therapy to achieve long-term continuous or controllable expression of biotherapeutics. Sci Adv. 2022 Jul 15;8(28):eabm1890. doi: 10.1126/sciadv.abm1890. Epub 2022 Jul 13.
Expression from AAV vector switched on by splice-targeted Vivo-Morpholino in HEK293 and in mice. "12.5 mg/kg...delivered intravenously via tail vein injection for up to two doses, given on two consecutive days for each treatment course."
Lu-Nguyen N, Dickson G, Malerba A, Popplewell L. Long-Term Systemic Treatment of a Mouse Model Displaying Chronic FSHD-like Pathology with Antisense Therapeutics That Inhibit DUX4 Expression. Biomedicines. 2022 Jul 7;10(7):1623. doi: 10.3390/biomedicines10071623.
"...10 mg/kg of either vivoPMO-PACS4 (PACS4) or vivoPMO-SCR (SCR) while the ACTA1-MCM mice received volume-matched saline, which was considered as the positive control (CTRL). Either vivoPMOs or saline was delivered one week after the second TMX dose and repeated once/twice per week thereafter for a total of 12 injections."
Rodríguez-Arrizabalaga M, Hernández-Núñez I, Candal E, Barreiro-Iglesias A. Use of vivo-morpholinos for gene knockdown in the postnatal shark retina. bioRxiv. 2022;[preprint] doi:10.1101/2022.07.11.499558
Scyliorhinus canicula (catshark) intraocular injection
Liu Y, Yang Y, Lin Y, Wei B, Hu X, Xu L, Zhang W, Lu J. N6 -methyladenosine-modified circRNA RERE modulates osteoarthritis by regulating β-catenin ubiquitination and degradation. Cell Prolif. 2022 Jun 22:e13297. doi: 10.1111/cpr.13297. Online ahead of print.
"...5 nmol of vivo-MO-circRere or control in 10 μL of sterile water was IA-injected into the affected knees of DMM mice (commencing 1 week after DMM surgery, and once weekly for 3 weeks)."
Guardado Rivas MO, Stuart SD, Thach D, Dahan M, Shorr R, Zachar Z, Bingham PM. Evidence for a novel, effective approach to targeting carcinoma catabolism exploiting the first-in-class, anti-cancer mitochondrial drug, CPI-613. PLoS ONE. 2022;17(6):e0269620. doi:10.1371/journal.pone.0269620
cell culture: AsPC-1
Vorlova S, Blahetek G, Barbieri R, Kerstan Y, Rosa A, Bundalo M, Egg M, Butt E, Henke E, Dölken L, Houben R, Schilling B, Fischer U, Erhard F, Zernecke A. U1 snRNP suppresses microRNA biogenesis by alternative intronic polyadenylation in melanoma. bioRxiv. 2022;[preprint] doi:10.1101/2022.06.01.479622
Ranasinghe N, Lin C-H, Lee T-H. Cholesterol Accumulation in Livers of Indian Medaka, Oryzias dancena, Acclimated to Fresh Water and Seawater. Front Mar Sci. 2022;9:{Epub] doi:10.3389/fmars.2022.891706
"Vivo-MO or control was gently injected into the region closer to the anal pore in the abdomen of the medaka"
Hall AE, Pohl SÖ, Cammareri P, Aitken S, Younger NT, Raponi M, Billard CV, Carrancio AB, Bastem A, Freile P, Haward F, Adams IR, Caceres JF, Preyzner P, von Kriegsheim A, Dunlop MG, Din FV, Myant KB. RNA splicing is a key mediator of tumour cell plasticity and a therapeutic vulnerability in colorectal cancer. Nat Commun. 2022 May 19;13(1):2791. doi: 10.1038/s41467-022-30489-z.
mouse intestinal cell culture
Tse KM, Vandenbon A, Cui X, Mino T, Uehata T, Yasuda K, Sato A, Tsujimura T, Hia F, Yoshinaga M, Kinoshita M, Okuno T, Takeuchi O. Enhancement of Regnase-1 expression with stem loop-targeting antisense oligonucleotides alleviates inflammatory diseases. Sci Transl Med. 2022 May 11;14(644):eabo2137. doi: 10.1126/scitranslmed.abo2137. Epub 2022 May 11.
Mouse intratracheal delivery and intracranial injection
Das SC, Althobaiti YS, Hammad AM, Alasmari F, Sari Y. Role of suppressing GLT-1 and xCT in ceftriaxone-induced attenuation of relapse-like alcohol drinking in alcohol-preferring rats. Addict Biol. 2022;[Epub] doi:10.1111/adb.13178
Tavakoli S, Zhu S, Matsudaira P. Cell Clusters Containing Intestinal Stem Cells Line the Zebrafish Intestine Intervillus Pocket. iScience. 2022;104280 [Epub]. doi:10.1016/j.isci.2022.104280
Delivery to zebrafish gut by intestinal gavage
Pan S, Cesarek M, Godoy C, Co CM, Schindler C, Padilla K, Haskell A, Barreda H, Story C, Poole R, Dabney A, Gregory CA. Morpholino-driven blockade of Dkk-1 in osteosarcoma inhibits bone damage and tumour expansion by multiple mechanisms. Br J Cancer. 2022;[Epub] doi:10.1038/s41416-022-01764-z
cell culture: (RFP)-labeled MOSJ-Dkk1 murine osteochondral sarcoma line, xenograft in Foxn1-/Foxn1- mice
Kobayashi Y, Chhoeu C, Li J, Price KS, Kiedrowski LA, Hutchins JL, Hardin AI, Wei Z, Hong F, Bahcall M, Gokhale PC, Jänne PA. Silent mutations reveal therapeutic vulnerability in RAS Q61 cancers. Nature. 2022 Mar 2. doi: 10.1038/s41586-022-04451-4. Online ahead of print.
Treatment of tumor xenografts in mice
Zhou H, Shen C, Guo Y, Huang X, Zheng B, Wu Y. The plasminogen receptor directs maintenance of spermatogonial stem cells by targeting BMI1. Mol Biol Rep. 2022 Feb 27. doi: 10.1007/s11033-022-07289-1. Online ahead of print.
murine explants
Haas M, Caron G, Chatonnet F, Manenti S, Alaterre E, Devin J, Delaloy C, Bertolin G, Viel R, Pignarre A, Llamas Gutierrez F, Marchalot A, Decaux O, Tarte K, Delpy L, Moreaux J, Fest T. Pivotal role of PIM2 kinase in plasmablast generation and plasma cell survival, opening new treatment options in myeloma. Blood. 2022 Feb 2:blood.2021014011. doi:10.1182/blood.2021014011. Online ahead of print.
cell culture: human B cells
De Gasperi R, Mo C, Azulai D, Wang Z, Harlow LM, Du Y, Graham Z, Pan J, Liu XH, Guo L, Zhang B, Ko F, Raczkowski AM, Bauman WA, Goulbourne CN, Zhao W, Brotto M, Cardozo CP. Numb is required for optimal contraction of skeletal muscle. J Cachexia Sarcopenia Muscle. 2022 Jan 9. doi: 10.1002/jcsm.12907. Online ahead of print.
cell culture: mouse primary myoblasts, myotubes
Bizen N, Bepari AK, Zhou L, Abe M, Sakimura K, Ono K, Takebayashi H. Ddx20, an Olig2 binding factor, governs the survival of neural and oligodendrocyte progenitor cells via proper Mdm2 splicing and p53 suppression. Cell Death Differ. 2022 Jan 1. doi: 10.1038/s41418-021-00915-8. Online ahead of print.
"1 μl of 1 μg/μl MO ... was injected into the ventricles of the embryonic mouse brains at E14.5. Nighty-six hours after injection, the brains were collected and used for immunohistochemistry or RT-qPCR."
Vavrušová Z, Chu DB, Nguyen A, Fish JL, Schneider RA. Differential regulation of SHH signaling and the developmental control of species-specific jaw size through neural crest-mediatedGas1 expression. bioRxiv. 2021;[preprint] doi:10.1101/2021.12.17.473230
Injection of Vivo-Morpholino in ovo to mandible of quail embryo
Raman R, Fallatah W, Al Qaryoute A, Ryon M, Jagadeeswaran P. Knockdown and Knockout of Tissue Factor Pathway Inhibitor in Zebrafish. Thromb Haemost. 2021 Dec 16. doi: 10.1055/a-1723-4075. Online ahead of print.
i.v. injection of Vivo-Morpholino-DNA piggyback hybrid into adult zebrafish
Liu Y, Yu X, Huang A, Zhang X, Wang Y, Geng W, Xu R, Li S, He H, Zheng B, Chen G, Xu Y. INTS7-ABCD3 Interaction Stimulates the Proliferation and Osteoblastic Differentiation of Mouse Bone Marrow Mesenchymal Stem Cells by Suppressing Oxidative Stress. Front Physiol. 2021;12:2004. doi:10.3389/fphys.2021.758607
Delivery into cultures of C57BL/6 murine bone marrow mesenchymal stem cells (BM-MSC)
Nugent BM, Stiver KA, Han J, Kindsvater HK, Marsh-Rollo SE, Hofmann HA, Alonzo SH. Morpholino-mediated knockdown of the brain mineralocorticoid receptor affects glucocorticoid signaling and neuroplasticity in wild ocellated wrasse (Symphodus ocellatus). bioRxiv. 2021;[preprint] doi:10.1101/2021.11.17.468986
Intracerebroventricular injection into the third ventricle of 1μl of 400nmol/μl Vivo-Morpholino over 30 seconds
Bhaskar M, Mukherjee S, Basu A. Involvement of RIG-I Pathway in Neurotropic Virus-Induced Acute Flaccid Paralysis and Subsequent Spinal Motor Neuron Death. mBio. 2021 Nov 16:e0271221. doi: 10.1128/mBio.02712-21. Online ahead of print.
10 day old BALB/c mice, intracranial injection of 18.5 mg/kg Vivo-Morpholino, i.p. viral challenge 24 hours later.
Wu N, Carpino G, Ceci L, Baiocchi L, Francis H, Kennedy L, Zhou T, Chen L, Sato K, Kyritsi K, Meadows V, Ekser B, Franchitto A, Mancinelli R, Onori P, Gaudio E, Glaser S, Alpini G. MT1 but not MT2 melatonin receptor knockout decreases biliary damage and liver fibrosis during cholestatic liver injury. Hepatology. 2021 Nov 7. doi: 10.1002/hep.32233. Online ahead of print.
mice
Chattopadhyay D, Das S, Guria S, Basu S, Mukherjee S. Fetuin-A regulates adipose tissue macrophage content and activation in insulin resistant mice through MCP-1 and iNOS: Involvement of IFNgamma-JAK2-STAT1 pathway. Biochem J. 2021 Nov 1:BCJ20210442. doi: 10.1042/BCJ20210442. Online ahead of print.
"In vivo morpholino oligos specific for FetA was injected dissolved in physiological saline (25 nM) for 5 days in a row to generate FetA knockdown mice model. ... On the 10th day following the final injection, the mice were sacrificed."
Marchalot A, Horiot C, Lambert J-M, Carrion C, Oblet C, Pollet J, Cogné M, Moreau J, Laffleur B, Delpy L. Targeting IgE polyadenylation signal with antisense oligonucleotides decreases IgE secretion and plasma cell viability. J Allergy Clin Immunol. 2021;[Epub ahead of print] doi:10.1016/j.jaci.2021.09.039
B cells, myeloma cells; InEps mouse
Matrone G, Jung SY, Choi JM, Jain A, Leung H-CE, Rajapakshe K, Coarfa C, Rodor J, Denvir MA, Baker AH, Cooke JP. Nuclear S-nitrosylation impacts tissue regeneration in zebrafish. Nat Comm. 2021;12(1):6282. doi:10.1038/s41467-021-26621-0
"Adult zebrafish were anesthetized in tricaine 0.05 mg/mL, and 2 μL of 0.1 mmol/L vivo-Mo solution, previously loaded in a glass capillary, was injected into the retro-orbital vein, as previously described, on days 10, 8, 6, 4, 2 and 0 before tailfin amputation."
Chiang K-Y, Li Y-W, Li Y-H, Huang S-J, Wu C-L, Gong H-Y, Wu J-L. Progranulin A Promotes Compensatory Hepatocyte Proliferation via HGF/c-Met Signaling after Partial Hepatectomy in Zebrafish. Int J Mol Sci. 2021;22(20):11217. doi:10.3390/ijms222011217
"Three‐month‐old adult Tg(fabp10:EGFP) zebrafish were administered intraperitoneal injections (IP‐injections) of 5 μL GrnA vivo‐MO at concentrations of 0.5, 0.25, and 0.125 mM (PBS as a control) ..., and the fish survival rate (n = 20 per group) was assessed every 12 h after injection. At 24 h after injection, the survival rate of the 0.5 mM group was 5%, and the 0.25 mM group survival rate was 45%. Fish treated with 0.125 mM exhibited the highest survival rate of all groups at 95% at 24 h post‐injection ... . Therefore, 0.125 mM GrnA vivo‐MO was selected for knockdown of GrnA expression in Tg(fabp10:EGFP). The injections were made every 24 h because the suppressive effect (0.39‐fold expression) was observed at 24 h, but the expression recovered at 48 h post‐injection (Supplementary Figure S1C). GrnA vivo‐MO injections every 24 h could maintain GrnA expression at levels between 0.46‐fold and 0.57‐fold until 96 h post‐injection."
Kruyer A, Dixon D, Angelis A, Amato D, Kalivas PW. Astrocytes in the ventral pallidum extinguish heroin seeking through GAT-3 upregulation and morphological plasticity at D1-MSN terminals. Mol Psychiatry. 2021;[Epub]. doi:10.1038/s41380-021-01333-5
Delivered though cannula 1.5 mm above the dorsolateral ventral pallidum (dlVP). "...rats received bilateral infusions (0.7 μL, 0.15 μL/min, 5 min diffusion) of an ezrin-targeted vivo-morpholino oligo ..., a GAT-3-targeted vivo-morpholino oligo ..., or a standard control oligo with no expected binding target in the brain ... under light isoflurane anesthesia (5% induction, 1.5% maintenance)."
Alsrhani A, Raman R, Jagadeeswaran P. Trypsin induces an aversive response in zebrafish by PAR2 activation in keratinocytes. PLoS One. 2021 Oct 8;16(10):e0257774. doi: 10.1371/journal.pone.0257774. eCollection 2021. br> Piggyback delivery: "5 dpf larvae were microinjected either with 4 nl aSO/cVMO or cSO/cVMO hybrids. Intravenous microinjection was made at the site where the inferior vena cava delivers blood to the yolk sac..."
Shiuchi T, Otsuka A, Shimizu N, Chikahisa S, Séi H. Feeding Rhythm-Induced Hypothalamic Agouti-Related Protein Elevation via Glucocorticoids Leads to Insulin Resistance in Skeletal Muscle. Int J Mol Sci. 2021;22(19):10831. doi:10.3390/ijms221910831
"Vivo-morpholinos were injected into the bilateral arcuate hypothalamus twice weekly"
Ivanova AS, Tereshina MB, Araslanova KR, Martynova NY, Zaraisky AG. The Secreted Protein Disulfide Isomerase Ag1 Lost by Ancestors of Poorly Regenerating Vertebrates Is Required for Xenopus laevis Tail Regeneration. Front Cell Dev Biol. 2021;9:738940. doi:10.3389/fcell.2021.738940
"...we injected fresh tail stumps by the anti-sense vivo-MOs..." "For better spreading of vivo-MO, we injected 0.4 mM solution in a mixture with the fluorescent tracer FLD into the notochord and both fins in the direction of tail growth near the amputated edge immediately after amputation. We repeated injections once per day during 1–4 dpa. After blastema formation, we injected the solution into the fins and notochord and the blastema."
Novak JS, Spathis R, Dang UJ, Fiorillo AA, Hindupur R, Tully CB, Mázala DAG, Canessa E, Brown KJ, Partridge TA, Hathout Y, Nagaraju K. Interrogation of Dystrophin and Dystroglycan Complex Protein Turnover After Exon Skipping Therapy. J Neuromuscul Dis. 2021 Sep 23. doi: 10.3233/JND-210696. Online ahead of print.
"Mice (treated mdx cohorts 2–6) were anesthetized using 4%isoflurane and PMO was administered PMO (I.V. tail vein, 28-guage/0.3 mL syringe) once daily for 3 days as follows: 12.5 mg/kg (day 0), 9.375 mg/kg (day 1), 6.25 mg/kg (day 2)."
Alharbi A, Iyer N, AlQaryoute A, Raman R, Burks D, Azad R, Jagadeeswaran P. Role of Ribosomal RNA Released from Red Cells in Blood Coagulation in Zebrafish and Humans. Blood Adv. 2021 Sep 21:bloodadvances.2020003325. doi: 10.1182/bloodadvances.2020003325. Online ahead of print.
Vivo-Morpholino piggyback delivering DNA
De Gasperi R, Mo C, Azulai D, Wang Z, Harlow LM, Du Y, Graham Z, Pan J, Liu X-H, Guo L, Zhang B, Ko F, Raczkowski AM, Bauman WA, Goulbourne CN, Zhao W, Brotto M, Cardozo CP. Numb is required for optimal contraction of skeletal muscle. bioRxiv. 2021;[preprint] doi:10.1101/2021.09.03.453960
cell culture: mouse primary myotubes
Biswas K, Couillard M, Cavallone L, Burkett S, Stauffer S, Martin BK, Southon E, Reid S, Plona TM, Baugher RN, Mellott SD, Pike KM, Albaugh ME, Maedler-Kron C, Hamel N, Tessarollo L, Marcus V, Foulkes WD, Sharan SK. A novel mouse model of PMS2 founder mutation that causes mismatch repair defect due to aberrant splicing. Cell Death Dis. 2021 Sep 6;12(9):838. doi: 10.1038/s41419-021-04130-8
Vivo-Morpholino targeting mutant splice site was not effective for mouse intestinal target.
Hildreth JEK, Moulton JD, Alcendor DJ. Vivo-Morpholino-Based Antiviral for SARS-CoV-2: Implications for Novel Therapies in the Treatment of Acute COVID-19 Disease. Biomedicines. 2021;9(8):1018. doi:10.3390/biomedicines9081018
Vero E6 cells
Ran C, Xie M, Li J, Xie Y, Ding Q, Li Y, Zhou W, Yang Y, Zhang Z, Olsen RE, Zhou Z. Dietary Nucleotides Alleviate Hepatic Lipid Deposition via Exogenous AMP-Mediated AMPK Activation in Zebrafish. J Nutr. 2021;nxab232 {Epub] doi:doi.org/10.1093/jn/nxab232
Raman R, Fallatah W, Al Qaryoute A, Dhinoja S, Jagadeeswaran P. Knockdown screening of chromatin binding and regulatory proteins in zebrafish identified Suz12b as a regulator of tfpia and an antithrombotic drug target. Sci Rep. 2021 Jul 27;11(1):15238. doi: 10.1038/s41598-021-94715-2.
Vivo-Morpholino piggyback hybrid with nucleic acid oligo used in adult zebrafish
Pan S, Cesarek M, Godoy C, Co C, Schindler C, Padilla K, Poole R, Dabney A, Gregory C. Morpholino-driven transcriptional blockade of Dkk-1 in experimental osteosarcoma inhibits bone damage and tumor expansion by multiple mechanisms. Res Sq. 2021;[preprint] doi:10.21203/rs.3.rs-677459/v1
Vivo-Morpholinos were administered at 12.5 mg/kg by intraperitoneal injection every 2 days.
Hu X, Gao S, Wang P, Zhou Y, Chen K, Chen Q, Wang B, Hu W, Cheng P, Eid R, Giraud-Panis MJ, Wang L, Gilson E, Ye J, Lu Y. The knockdown efficiency of telomere associated genes with specific methodology in a zebrafish cell line. Biochimie. 2021 Jun 29:S0300-9084(21)00158-9. doi: 10.1016/j.biochi.2021.06.013. Online ahead of print.
Hamdani EH, Popek M, Frontczak-Baniewicz M, Utheim TP, Albrecht J, Zielińska M, Chaudhry FA. Perturbation of astroglial Slc38 glutamine transporters by NH4+ contributes to neurophysiologic manifestations in acute liver failure. FASEB J. 2021;35:e21588. doi:10.1096/fj.202001712RR
Vivo-Morpholino in osmotic pump to frontal cortex of C57Bl6 mouse
Iyer N, Al Qaryoute A, Kacham M, Jagadeeswaran P. Identification of Zebrafish Ortholog for Human Coagulation Factor IX and its Age-dependent Expression. J Thromb Haemost. 2021 May 11. doi: 10.1111/jth.15365. Online ahead of print.
Piggyback hybrids, Vivo-Morpholino carrying DNA for RNase-H activity
Gudiño V, Pohl SÖ, Billard CV, Cammareri P, Bolado A, Aitken S, Stevenson D, Hall AE, Agostino M, Cassidy J, Nixon C, von Kriegsheim A, Freile P, Popplewell L, Dickson G, Murphy L, Wheeler A, Dunlop M, Din F, Strathdee D, Sansom OJ, Myant KB. RAC1B modulates intestinal tumourigenesis via modulation of WNT and EGFR signalling pathways. Nat Commun. 2021 Apr 20;12(1):2335. doi:10.1038/s41467-021-22531-3.
cell culture: CMT93 cells; human organoids
Roy JCL, Vitalo A, Andrew MA, Mota-Silva E, Kovalenko M, Burch Z, Nhu AM, Cohen PE, Grabczyk E, Wheeler VC, Mouro Pinto R. Somatic CAG expansion in Huntington's disease is dependent on the MLH3 endonuclease domain, which can be excluded via splice redirection. Nucleic Acids Res. 2021 Mar 22:gkab152. doi:10.1093/nar/gkab152. Online ahead of print.
25 mg/kg tail vein injection; treatment started in 4-week-old mice, injected 3x/week, stopped at 12 weeks age
Petersen I, Chang WWJ, Hu MY. Na+/H+-exchangers differentially contribute to midgut fluid sodium and proton concentration in the sea urchin larva. J Exp Biol. 2021;jeb.240705 doi: 10.1242/jeb.240705 Published 5 March 2021
Chandrakar P, Seth A, Rani A, Dutta M, Parmar N, Descoteaux A, Kar S. Jagged–Notch-mediated divergence of immune cell crosstalk maintains the anti-inflammatory response in visceral leishmaniasis. J Cell Sci. 2021;134:jcs252494 doi:10.1242/jcs.252494
Balb/c mice with Leishmania donovani infections, 5mg/kg Vivo-MO given twice/week for six weeks
Guo R, Ge K, Wang Y, Lu M, Li F, Tian L, Gan L, Sheng D. LIM Homeobox 4 (lhx4) regulates retinal neural differentiation and visual function in zebrafish. Sci Rep. 2021;11:1977. doi:10.1038/s41598-021-81211-w
Injected into one eye of embryos at 26–27 hpf
Lu-Nguyen N, Malerba A, Dickson G, Popplewell L. Systemic antisense therapeutics inhibiting DUX4 expression improves muscle function in an FSHD mouse model. bioRxiv. 2021;[preprint] doi:10.1101/2021.01.14.426659
Targeting polyadenylation signal and cleavage site on DUX4 3'-UTR; Multiple doses of Vivo-Morpholinos in mice at 10 mg/kg, i.p. injection
Tsata V, Möllmert S, Schweitzer C, Kolb J, Möckel C, Böhm B, Rosso G, Lange C, Lesche M, Hammer J, Kesavan G, Beis D, Guck J, Brand M, Wehner D. A switch in pdgfrb+ cell-derived ECM composition prevents inhibitory scarring and promotes axon regeneration in the zebrafish spinal cord. Dev Cell. 2021;[Epub ahead of print] doi:10.1016/j.devcel.2020.12.009
Injection mix containing 0.475 mM Vivo-MO microinjected as 10 repetitive injections of 1 nl each into the pericardial vein of 3 dpf larvae.
Gao W, Huang Q, Qin T, Mu H, Long F, Ren L, Tang H, Liu J, Zeng M. HSP60 regulates monosodium urate crystal-induced inflammation by activating the TLR4-NF- κ B-MyD88 signaling pathway and disrupting mitochondrial function. Oxid Med Cell Longev. 2020;[Epub] doi:10.1155/2020/8706898
Vivo-Morpholino injected into mouse footpad at 15mg/(kg body weight)
Gao T, Lin M, Wu Y, Li K, Liu C, Zhou Q, Shen C, Zheng B, Huang X. Transferrin receptor (TFRC) is essential for meiotic progression during mouse spermatogenesis. Zygote. 2020 Dec 16:1-7. doi: 10.1017/S0967199420000659. Online ahead of print.
Delivery into mouse testis explants
Ratliff ML, Shankar M, Guthridge JM, James JA, Webb CF. TLR engagement induces ARID3a in human blood hematopoietic progenitors and modulates IFNalpha production. Cell Immunol. 2020 Nov;357:104201. doi: 10.1016/j.cellimm.2020.104201. Epub 2020 Sep 9.
Delivery to human hematopoietic stem progenitor cell culture (HSPCs)
Boender AJ, Bontempi L, Nava L, Pelloux Y, Tonini R. Striatal astrocytes shape behavioral flexibility via regulation of the glutamate transporter EAAT2. Biol Psychiatry. 2020;[Epub ahead of print] doi:10.1016/j.biopsych.2020.11.015
mice
Raman R, Ryon M, Jagadeeswaran P. RNaseH-mediated simultaneous piggyback knockdown of multiple genes in adult zebrafish. Sci Rep. 2020;10:20187 doi:10.1038/s41598-020-76655-5
Wu CS, Lu YF, Liu YH, Huang CJ, Hwang SL. Zebrafish Cdx1b modulates epithalamic asymmetry by regulating ndr2 and lft1 expression. Dev Biol. 2020 Nov 13:S0012-1606(20)30293-1. doi: 10.1016/j.ydbio.2020.11.001. Online ahead of print.
Injection into 18hpf zebrafish forebrain ventricle
Khandekar G, Iyer N, Jagadeeswaran P. Prostasin and hepatocyte growth factor B in factor VIIa generation: Serine protease knockdowns in zebrafish. Res Pract Thromb Haemost. 2020 Sep 22;4(7):1150-1157. doi: 10.1002/rth2.12428. eCollection 2020 Oct.
Vivo-Morpholino carries partially-complementary DNA oligo into cells after iv injections into adult zebrafish, second injection 24 hours later.
Chrisafis G, Wang T, Moissoglu K, Gasparski AN, Ng Y, Weigert R, Lockett SJ, Mili S. Collective cancer cell invasion requires RNA accumulation at the invasive front. Proc Natl Acad Sci U S A. 2020;[Epub ahead of print] doi:10.1073/pnas.2010872117
Cell culture: MDA-MB-231 speroids
Li Y, Gao X, Wei C, Guo R, Xu H, Bai Z, Zhou J, Zhu J, Wang W, Wu Y, Li J, Zhang Z, Xie X. Modification of Mcl-1 alternative splicing induces apoptosis and suppresses tumor proliferation in gastric cancer. Aging. 2020;12. doi:10.18632/aging.103766
"Mice were grouped after tumor formation and then injected with vivo-morpholino-modified Mcl-1-specific SBOs or a non-targeting vivo-morpholino-modified oligonucleotide (as the control) ... by local multipoint administration. Dosing (1.25 or 6.25 mg/kg SBOs) was repeated daily for 3 days, and the mice were sacrificed on day 4."
Duque-Wilckens N, Torres LY, Yokoyama S, Minie VA, Tran AM, Petkova SP, Hao R, Ramos-Maciel S, Rios RA, Jackson K, Flores-Ramirez FJ, Garcia-Carachure I, Pesavento PA, Iñiguez SD, Grinevich V, Trainor BC. Extrahypothalamic oxytocin neurons drive stress-induced social vigilance and avoidance. Proc Natl Acad Sci U S A. 2020 Oct 5:202011890. doi: 10.1073/pnas.2011890117. Online ahead of print.
Peromyscus californicus (California mice), medioventral bed nucleus of the stria terminalis (BNSTmv) Vivo-Morpholino injection
Marchalot A, Lambert J-M, Boyer F, Pollet J, Moreau J, Feuillard J, Faumont N, Delpy L. Splice switching oligonucleotide mediated gene knockdown in B cells and plasma cells. bioRxiv. 2020;[preprint] doi:10.1101/2020.09.18.302984
SUDHL4 and OCILY10 (DLBCL) lines, primary Human Peripheral Blood Mononuclear B Cells (PBMC)
Howard E, Lewis G, Galle-Treger L, Hurrell BP, Helou DG, Shafiei-Jahani P, Painter J, Muench GA, Soroosh P, Akbari O. IL-10 production by ILC2s requires Blimp-1 and cMaf, modulates cellular metabolism and ameliorates airway hyperreactivity. J Allergy Clin Immunol. 2020 Sep 6:S0091-6749(20)31234-3. doi: 10.1016/j.jaci.2020.08.024. Online ahead of print.
Cultured Murine Group 2 Innate Lymphoid Cells (ILC2); "2.5 μM ... in-vivo morpholinos ... were added as free uptake oligos 24 h before rmIL-4 (10ng/ml) addition."
Popek M, Bobula B, Sowa J, Hess G, Frontczak-Baniewicz M, Albrecht J, Zielińska M. Physiology and morphological correlates of excitatory transmission are preserved in glutamine transporter SN1- depleted mouse frontal cortex. Neurosci. 2020;[Epub] doi:10.1016/j.neuroscience.2020.08.019
Derenne A, Tassin A, Nguyen TH, De Roeck E, Jenart V, Ansseau E, Belayew A, Coppée F, Declèves A-E, Legrand A. Induction of a local muscular dystrophy using electroporation in vivo: an easy tool for screening therapeutics. Sci Rep. 2020;10(1):11301. doi:10.1038/s41598-020-68135-7
mouse i.p. injection
Marcheva B, Perelis M, Weidemann BJ, Taguchi A, Lin H, Omura C, Kobayashi Y, Newman MV, Wyatt EJ, McNally EM, Fox JEM, Hong H, Shankar A, Wheeler EC, Ramsey KM, MacDonald PE, Yeo GW, Bass J. A role for alternative splicing in circadian control of exocytosis and glucose homeostasis. Genes Dev. 2020 Jul 2. doi: 10.1101/gad.338178.120. Online ahead of print.
Delivery into cultured explants: murine pancreatic islets
Li L, Hobson L, Perry L, Clark B, Heavey S, Haider A, Sridhar A, Shaw G, Kelly J, Freeman A, Wilson I, Whitaker H, Nurmemmedov E, Oltean S, Porazinski S, Ladomery M. Targeting the ERG oncogene with splice-switching oligonucleotides as a novel therapeutic strategy in prostate cancer. Br J Cancer. 2020;[Epub ahead of print]doi:10.1038/s41416-020-0951-2. Online ahead of print.
Cell culture MG63, VCaP; xenograft mice; primary human tumor explant. Vivo-Morpholinos used with Endo-Porter in VCaP cells.
Lentz JJ, Hathaway M, Robillard KN, Ponnath A, Fisher M, Rigo F, Alapure BV. Optimizing oligotherapeutics for visual loss in Usher syndrome mice. Invest Ophthalmol Vis Sci. 2020;Vol.61:2445.
mice intravitreal injection
Raktoe RS, Rietveld MH, Out-Luiting JJ, Kruithof-de Julio M, van Zuijlen PPM, van Doorn R, El Ghalbzouri A. Exon skipping of TGFβRI affects signalling and ECM expression in hypertrophic scar-derived fibroblasts. Scars Burn. Heal. 2020;6;[Epub] doi:10.1177/2059513120908857
cell culture: primary human fibroblasts
Duque-Wilckens N, Torres L, Yokoyama S, Minie V, Tran A, Petkova S, Hao R, Ramos-Maciel S, Rioa R, Jackson K, Flores-Ramirez F, Garcia-Carachure I, Pesavento PA, Iniguez S, Grinevich V, Trainor BC. Extra-hypothalamic oxytocin neurons drive stress-induced social vigilance and avoidance. bioRxiv. 2020;[preprint] doi:10.1101/2020.06.02.129981 "...animals were randomly assigned to receive one bilateral 0.2uL injection of 100pM morpholino missense or antisense aimed a little anterior to medioventral BNST (BNSTmv, AP:0.39, ML: 1.1, DV:-5.85)."
Lorenson MY, Chen KE, Walker AM. Enzyme-linked oligonucleotide hybridization assay for direct oligo measurement in blood. Biol Methods Protoc. 2019 Jan 7;4(1):bpy014. doi: 10.1093/biomethods/bpy014. eCollection 2019.
Kyritsi K, Francis H, Zhou T, Ceci L, Wu N, Yhang Z, Meng F, Chen L, Baiocchi L, Kundu D, Kennedy L, Liangpunsakul S, Wu C, Glaser S, Alpini G. Downregulation of p16 decreases biliary damage and liver fibrosis in the Mdr2-/- mouse model of primary sclerosing cholangitis. Gene Expr. 2020 May 11. doi: 10.3727/105221620X15889714507961. [Epub ahead of print]
Marchelot A, Ashi MO, Lambert JM, Carrion C, Lecardeur S, Srour N, Delpy L, Le Pennec S. Uncoupling Splicing From Transcription Using Antisense Oligonucleotides Reveals a Dual Role for I Exon Donor Splice Sites in Antibody Class Switching. Front Immunol. 2020;11:780. doi:10.3389/fimmu.2020.00780
cell culture: murine splenic B cells
Chen JL, Zhang P, Abe M, Aikawa H, Zhang L, Frank AJ, Zembryski T, Hubbs C, Park HJ, Withka J, Steppan C, Rogers L, Cabral S, Pettersson M, Wager TT, Fountain MA, Rumbaugh G, Childs-Disney JL, Disney MD. Design, Optimization, and Study of Small Molecules That Target Tau Pre-mRNA and Affect Splicing. J Am Chem Soc. 2020;[Epub ahead of print] doi:10.1021/jacs.0c00768
cell culture: HeLa
Gao W, Huang Q, Qin T, Mu H, Long F, Ren L, Tang H, Liu J, Zeng M. HSP60 regulates monosodium urate crystal-induced inflammation by activating the TLR4-NF- κ B-MyD88 signaling pathway and disrupting mitochondrial function. Research Square. 2020;[preprint] doi:10.21203/rs.3.rs-25061/v1
mice, 15mg/kg i.p. injection or 15mg/kg foot pad or ankle joint injection
Zhang J, Wang Y, Li S-Q, Fang L, Wang X-Z, Li J, Zhang H-B, Huang B, Xu Y-M, Yang W-M, Lin J, Min Q-H, ZLiao Z-H, Wu Y, Liu J. Correction of Bcl‐x splicing improves responses to imatinib in chronic myeloid leukaemia cells and mouse models. Brit J Hematol. 2020;[Epub] doi:10.1111/bjh.16472
Stratman AN, Farrelly OM, Mikelis CM, Miller MF, Wang Z,Pham VN, Davis AE, Burns MC, Pezoa SA, Castranova D, Yano JJ, Kilts TM, Davis GE, Gutkind JS, Weinstein BM. Anti-angiogenic effects of VEGF stimulation on endothelium deficient in phosphoinositide recycling. Nat Comm. 2020;11(1):1204 doi:10.1038/s41467-020-14956-z
Vivo-Morpholinos "injected through the tail vein or retro-orbitally at a dose of 40 μl of 2000 nmol stock per mouse per day. vMO were stored at room temperature and heated at 65 °C for 15 min prior to use."
Kennedy L, Meadows V, Kyritsi K, Pham L, Kundu D, Kulkarni R, Cerritos K, Demieville J, Hargrove L, Glaser S, Zhou T, Jaeger V, Alpini G, Francis H. Amelioration of Large Bile Duct Damage by Histamine-2 Receptor Vivo-Morpholino Treatment. Am J Pathol. 2020;190(5):1018-29 doi:10.1016/j.ajpath.2020.01.013
Pasteuning-Vuhman S, Boertje-van der Meulen JW, Putten MV, Overzier M, Ten Dijke P, Kielbasa SM, Arindrarto W, Wolterbeek R, Lezhnina KV, Ozerov IV, Aliper AM, Hoogaars WM, Aartsma-Rus A, Loomans CJM. New function of the myostatin/activin type I receptor (ALK4) as a mediator of muscle atrophy and muscle regeneration. FASEB J. 2017 Jan;31(1):238-255. doi: 10.1096/fj.201600675r.
cell culture: C2C12 myoblasts, 4-wk-old male mdx or 3-mo-old male C57BL/6Jico WT mice
Injected with 40 μg ALK4 ViM or control nontargeting ViM at the same dose intramuscularly on 2 consecutive days. Mice were euthanized 1 wk (short-term treatment) or 7 wk (long-term treatment) after the first injection. Long-term treatment included an extra injection 4 wk after the first injection.
Hammond-Weinberger DR, Wang Y, Glavis-Bloom A, Spitzer NC. Mechanism for neurotransmitter-receptor matching. Proc Nat Acad Sci USA. 2020;[Epub ahead of print] doi:10.1073/pnas.1916600117
cell culture and Xenopus
Tanaka I, Chakraborty A, Saulnier O, Benoit-Pilven C, Vacher S, Labiod D, Lam EWF, Bièche I, Delattre O, Pouzoulet F, Auboeuf D, Vagner S, Dutertre M. ZRANB2 and SYF2-mediated splicing programs converging on ECT2 are involved in breast cancer cell resistance to doxorubicin. Nucleic Acids Res. 2020 Jan 16. pii: gkz1213. doi: 10.1093/nar/gkz1213. [Epub ahead of print]
Female Swiss Nu/nu mice intratumoral Vivo-Morpholino
Linker KE, Elabd MG, Tawadrous P, Cano M, Green KN, Wood MA, Leslie FM. Microglial activation increases cocaine self-administration following adolescent nicotine exposure. Nat Commun. 2020 Jan 16;11(1):306. doi: 10.1038/s41467-019-14173-3. Sprague-Dawley rat, intracranial
Jones M, Bellusci S. Imaging and Analysis of Mouse Embryonic Whole Lung, Isolated Tissue, and Lineage-Labelled Cell Culture. In: Bertoncello, I. (eds) Mouse Cell Culture. Methods in Molecular Biology. 2019;1940:109-27. Humana Press, New York, NY. doi:10.1007/978-1-4939-9086-3_8.
Zhong G, Wang H, He W, Li Y, Mou H, Tickner ZJ, Tran MH, Ou T, Yin Y, Diao H, Farzan M. A reversible RNA on-switch that controls gene expression of AAV-delivered therapeutics in vivo. Nat Biotechnol. 2019;[Epub ahead print] doi:10.1038/s41587-019-0357-y
Vivo-Morpholino used to control activity of an engineered ribozyme
Suhail A, Rizvi ZA, Mujagond P, Ali SA, Gaur P, Singh M, Ahuja V, Awasthi A, Srikanth CV. DeSUMOylase SENP7-Mediated Epithelial Signaling Triggers Intestinal Inflammation via Expansion of Gamma-Delta T Cells. Cell Rep. 2019 Dec 10;29(11):3522-3538.e7. doi: 10.1016/j.celrep.2019.11.028.
"Two doses of intraperitoneal injections were given on the first day and fourth day during the course of DSS treatment at the concentration of 10 mg/kg (20nM/mice)."
Marchalot A, Ashi MO, Lambert J-M, Srour N, Delpy L, Le Pennec S. Uncoupling splicing from transcription using antisense oligonucleotides reveals a dual role for I exon donor splice sites in antibody class switching. bioRxiv. 2019;[preprint] doi:10.1101/850867
"Stimulated splenic B cells were cultured in the presence of 2 μM ASO. Culture samples were harvested at day 2, 3 or 4 for subsequent flow cytometry analysis, ChIP assays or RNA extraction".
de Morree A, Klein JDD, Gan Q, Farup J, Urtasun A, Kanugovi A, Bilen B, van Velthoven CTJ, Quarta M, Rando TA. Alternative polyadenylation of Pax3 controls muscle stem cell fate and muscle function. Science. 2019 Nov 8;366(6466):734-738. doi: 10.1126/science.aax1694.
Demonbreun AR, Wyatt EJ, Fallon KS, Oosterbaan CC, Page PG, Hadhazy M, Quattrocelli M, Barefield DY, McNally EM. A gene-edited mouse model of limb-girdle muscular dystrophy 2C for testing exon skipping. Dis Model Mech. 2019 Nov 4;13(2). pii: dmm040832. doi: 10.1242/dmm.040832.
Evans BC, Fletcher RB, Kilchrist KV, Dailing EA, Mukalel AJ, Colazo JM, Oliver M, Cheung-Flynn J, Brophy CM, Tierney JW, Isenberg JS, Hankenson KD, Ghimire K, Lander C, Gersbach CA, Duvall CL. An anionic, endosome-escaping polymer to potentiate intracellular delivery of cationic peptides, biomacromolecules, and nanoparticles. Nat Comm. 2019;10:5012. doi:10.1038/s41467-019-12906-y
Hazra B, Chakraborty S, Bhaskar M, Mukherjee S, Mahadevan A, Basu A. miR-301a Regulates Inflammatory Response to Japanese Encephalitis Virus Infection via Suppression of NKRF Activity. J Immunol. 2019;[Epub ahead of pint] doi:10.4049/jimmunol.1900003
Vik Meadows V, Kennedy L, Hargrove L, Demieville J, Meng F, Virani S, Reinhart E, Kyritsi K, Invernizzi P, Yang Z, Wu N, Liangpunsakul S, Alpini G, Francis H. Downregulation of hepatic stem cell factor by Vivo-Morpholino treatment inhibits mast cell migration and decreases biliary damage/senescence and liver fibrosis in Mdr2−/− mice. Biochem Biophys Acta. 2019;[Epub ahead of print] doi:10.1016/j.bbadis.2019.165557
Zhou T, Kyritsi K, Wu N, Francis H, Yang Z, Chen L, O'Brien A, Kennedy L, Ceci L, Meadows V, Kusumanchi P, Wu C, Baiocchi L, Skill NJ, Saxena R, Sybenga A, Xie L, Liangpunsakul S, Meng F, Alpini G, Glaser S. Knockdown of vimentin reduces mesenchymal phenotype of cholangiocytes in the Mdr2−/− mouse model of primary sclerosing cholangitis (PSC). EBioMedicine, 2019;[in press] doi:10.1016/j.ebiom.2019.09.013
"...two tail vein injections for 1 week (30 mg/kg BW)"
Negri A, Ferrari M, Nodari R, Coppa E, Mastrantonio V, Zanzani S, Porretta D, Bandi C, Urbanelli S, Epis S. Gene silencing through RNAi and antisense Vivo-Morpholino increases the efficacy of pyrethroids on larvae of Anopheles stephensi. Malar J. 2019;18:294. doi:10.1186/s12936-019-2925-5
Bathing larvae in aqueous sol'n of Vivo-Morpholino
Zang L, Shimada Y, Nakayama H, Chen W, Okamoto A, Koide H, Oku N, Dewa T, Shiota M, Nishimura N. Therapeutic Silencing of Centromere Protein X Ameliorates Hyperglycemia in Zebrafish and Mouse Models of Type 2 Diabetes Mellitus. Front Genet. 2019;10:693. doi:10.3389/fgene.2019.00693
Zebrafish: "MO was intraperitoneally injected at a dose of 15 mg/kg fish weight twice a week during the 8-week experiment period."
Zhang Z, Ran C, Ding Q-w, Liu H-l, Xie M-x, Yang Y-l, Ya-dong Xie Y-d, Gao C-c, Zhang H-l, Zhou Z-g. Ability of prebiotic polysaccharides to activate a HIF1α-antimicrobial peptide axis determines liver injury risk in zebrafish. Comm Biol. 2019;2(1):274. doi:10.1038/s42003-019-0526-z
Uptake into zebrafish gut from Vivo-Morpholino in the surrounding water: "Embryos and larvae were reared in embryo medium at 28 °C to 4 day-postfertilization (dpf). Zebrafish larvae at 4 dpf were allocated randomly to tanks with 100 mL of water and 80 larvae per tank." "When applying the MO, zebrafish husbandry and all experimental procedures were the same as earlier described, except for gnotobiotic zebrafish medium that contained 100 nmol of tlr4ba MO, or standard control MO during the entire 1-week treatment"
Kruyer A, Scofield MD, Wood D, Reissner KJ, Kalivas PW. Heroin Cue-Evoked Astrocytic Structural Plasticity at Nucleus Accumbens Synapses Inhibits Heroin Seeking. Biol Psych. 2019;[Epub ahead of print] doi:10.1016/j.biopsych.2019.06.026
Male Sprague Dawley rats received bilateral NAcore brain infusion with Vivo-Morpholino through cannulae.
Chen J, Yu T, He X, Fu Y, Dai L, Wang B, Wu Y, He J, Li Y, Zhang F, Zhao J, Liu C. Dual roles of hydrogen peroxide in promoting zebrafish renal repair and regeneration. Biochem Biophys Res Commun. 2019 Jun 24. pii: S0006-291X(19)31178-7. doi: 10.1016/j.bbrc.2019.06.052. [Epub ahead of print]
zebrafish
Mukherjee S, Akbar I, Kumari B, Vrati S, Basu A, Banerjee A. Japanese Encephalitis Virus-induced let-7a/b interacted with the NOTCH-TLR7 pathway in microglia and facilitated neuronal death via caspase activation. J Neurochem. 2018 Dec 17. doi: 10.1111/jnc.14645. [Epub ahead of print]
Let7 miRNA knockdown with Vivo-Morpholino in mouse brain
Cho S, Vashisth M, Abbas A, Majkutv S, Vogel K, Xia Y, Ivanovska IL, Irianto J, Tewari M, Zhu K, Tichy ED, Mourkioti F, Tang H-Y, Greenberg R, Prosser BL, Discher DE. Mechanosensing by the lamina protects against nuclear rupture, DNA damage, and cell cycle arrest. bioRxiv. 2019;[preprint] doi:10.1101/583179
organ culture: Gallus gallus (chick) hearts
Liang Y, Rathnayake D, Huang S, Pathirana A, Xu Q, Zhang S. BMP signaling is required for amphioxus tail regeneration. Development. 2019 Jan 29. pii: dev.166017. doi:10.1242/dev.166017. [Epub ahead of print]
Delivery in Branchiostoma japonicum
Pantazis CB, James MH, Bentzley BS, Aston-Jones G. The number of lateral hypothalamus orexin/hypocretin neurons contributes to individual differences in cocaine demand. bioRxiv. 2019;[Preprint] doi:10.1101/547836
150nmol/0.3 uL Vivo-Morpholino in 0.5mM phosphate buffer infused over 2 minutes through cannula 2 mm dorsal to lateral hypothalamus in male Sprague-Dawley rats.
Ofer L, Dean MN, Zaslansky P, Kult S, Shwartz Y, Zaretsky J, Griess-Fishheimer S, Monsonego-Ornan E, Zelzer E, Shahar R. A novel nonosteocytic regulatory mechanism of bone modeling. PLoS Biol. 2019 Feb 1;17(2):e3000140. doi: 10.1371/journal.pbio.3000140. [Epub ahead of print]
Two μl of Vivo-Morpholino was injected into adult medaka to the ventral side of the fourth caudal vertebra. Injections were made every three days for two and a half weeks. Similar shorter term and lower dose experiments were also done.
Yin Y, Xu L, Chang Y, Zeng T, Chen X, Wang A, Groth J, Foo WC, Liang C, Hu H, Huang J. N-Myc promotes therapeutic resistance development of neuroendocrine prostate cancer by differentially regulating miR-421/ATM pathway. Mol Cancer. 2019 Jan 18;18(1):11. doi: 10.1186/s12943-019-0941-2.
Vivo-Morpholino targeting miR-421
Ko S-H, Liau Y-J, Chi Y-H, Lai M-J, Chiang Y-P, Lu C-Y, Chang L-Y, Tarn W-Y, Huang L-M. Interference of DNAJB6/MRJ Isoform Switch by Morpholino Inhibits Replication of HIV-1 and RSV. Molec Ther Nucl Acids. 2018;[Epub] doi:10.1016/j.omtn.2018.12.001
cell culture: HEK293T, THP-1, & Hep2, treated "in serum-free medium for 24 h. "
Hu N, Antoury L, Baran TM, Mitra S, Bennett CF, Rigo F, Foster TH, Wheeler TM. Non-invasive monitoring of alternative splicing outcomes to identify candidate therapies for myotonic dystrophy type 1. Nat Commun. 2018 Dec 7;9(1):5227. doi: 10.1038/s41467-018-07517-y. br> "...injected gastrocnemius muscles of TR;HSALR bi-transgenic mice with saline or 20 μg of a dendrimer-modified CAG repeat morpholino ASO (CAG25)"
Phumesin P, Junking M, Panya A, Yongpitakwattana P, Noisakran S, Limjindaporn T, Yenchitsomanus PT. Inhibition of dengue virus replication in monocyte-derived dendritic cells by vivo-morpholino oligomers. Virus Res. 2018 Nov 29. pii: S0168-1702(18)30380-0. doi: 10.1016/j.virusres.2018.11.014. [Epub ahead of print]
cell culture: human monocyte-derived dendritic cells
Ramsbottom SA, Molinari E, Srivastava S, Silberman F, Henry C, Alkanderi S, Devlin LA, White K, Steel DH, Saunier S, Miles CG, Sayer JA. Targeted exon skipping of a CEP290 mutation rescues Joubert syndrome phenotypes in vitro and in a murine model. Proc Natl Acad Sci U S A. 2018 Nov 16. pii: 201809432. doi: 10.1073/pnas.1809432115. [Epub ahead of print]
Vivo-Morpholinos were "injected into the tail vein of 28 day old Cep290Gt/Gt animals at a dose of 12.5 mg/Kg. 3 subsequent doses were administered at 3-4 day intervals over a period of 10 days, and tissues were collected 24 h after the final dose"
Norambuena A, Wallrabe H, Cao R, Wang DB, Silva A, Svindrych Z, Periasamy A, Hu S, Tanzi RE, Kim DY, Bloom GS. A novel lysosome-to-mitochondria signaling pathway disrupted by amyloid-β oligomers. EMBO J. 2018 Oct 22. pii: e100241. doi: 10.15252/embj.2018100241. [Epub ahead of print]
Vivo-Morpholino delivery to mouse cortical neurons
Zhao Y, Yang Z, Wang Y, Luo Y, Da F, Tao W, Zhou L, Wang D, Wei J. Both Gfrα1a and 1b are involved in the self-renewal and maintenance of spermatogonial stem cells in medaka. Stem Cells Dev. 2018 Oct 15. doi: 10.1089/scd.2018.0177. [Epub ahead of print]
Vivo-Morpholino delivery in SG3 cells (spermatogonial stem cell line) and in testicular organ culture
Huang K, Dang Y, Zhang P, Shen C, Sui X, Xia G, Qin Y, Jiao X, Wang C, Huo R, Chen Z-J. CAV1 regulates primordial follicle formation via the Notch2 signalling pathway and is associated with premature ovarian insufficiency in humans. Hum Reprod. 2018;dey299. doi:10.1093/humrep/dey299
Delivery into mouse ovaries
Jumbe SL, Porazinski SR, Oltean S, Mansell JP, Vahabi B, Wilson ID, Ladomery MR. The Evolutionarily Conserved Cassette Exon 7b Drives ERG's Oncogenic Properties. Transl Oncol. 2018;12(1):134-42. doi:10.1016/j.tranon.2018.09.001
Delivery into MG63 ( human osteoblast-like osteosarcoma) and VCaP (human prostate cancer vertebral metastasis) cultures.
Lykins JD, Filippova EV, Halavaty AS, Minasov G, Zhou Y, Dubrovska I, Flores KJ, Ludmilla A Shuvalova LA, Ruan J, El Bissati K, Dovgin S, Roberts CW, Woods S, Moulton JD, Moulton H, McPhillie M, Muench SP, Fishwick CWG, Sabini E, Shanmugam D, Roos DS, McLeod R, Anderson WF, Ngô H. CSGID Solves Structures and Identifies Phenotypes for Five Enzymes in Toxoplasma gondii. Front Cell Infect Microbiol. 2018;[Epub] doi:10.3389/fcimb.2018.00352
Vivo-Morpholino entered cultured human foreskin fibroblasts and then into the tachyzoites they contained. "HFFs were infected with YFP-expressing T. gondii tachyzoites and treated with vivoPMOs targeted against all five enzymes of interest. When treated at 10 μM concentrations, each morpholino resulted in approximately 50% reduction in fluorescence in vitro (between 44% for PGM-targeted vivoPMO to 56% reduction for OAT-targeted vivoPMO)."
McMillin M, Grant S, Frampton G, Petrescu AD, Kain J, Williams E, Haines R, Canady L, DeMorrow S. FXR-Mediated Cortical Cholesterol Accumulation Contributes to the Pathogenesis of Type A Hepatic Encephalopathy. Cell Mol Gastroenterol Hepatol. 2018 Mar 6;6(1):47-63. doi: 10.1016/j.jcmgh.2018.02.008. eCollection 2018.
"C57Bl/6 mice underwent surgery to implant Alzet brain infusion cannulas coupled to subcutaneous implanted minipumps (Alzet, Cupertino, CA) to directly infuse FXR morpholino or FXR mismatch (1 mg/kg) made up in a 10 μmol/L Endo-Porter solution to knock down FXR expression in the brain, which effectively knocked down the expression of FXR in neurons located adjacent to the injection site shown and characterized in previous studies." Vivo-Morpholino & Endo-Porter were loaded into the Alzet pump.
Oliveira AN, Hood DA. Effect of Tim23 Knockdown in vivo on Mitochondrial Protein Import and Retrograde Signaling to the UPRmt in Muscle. Am J Physiol Cell Physiol. 2018 Jun 27. doi: 10.1152/ajpcell.00275.2017. [Epub ahead of print]
Swaroop S, Mahadevan A, Shankar SK, Adlakha YK, Basu A. HSP60 critically regulates endogenous IL-1β production in activated microglia by stimulating NLRP3 inflammasome pathway. J Neuroinflammation. 2018 Jun 9;15(1):177. doi: 10.1186/s12974-018-1214-5.
"Among these, groups (v) and (vi) were intracranially injected with HSP60 vivo-morpholino at P8 (15 mg/kg of body weight of mice), while groups (iii) and (iv) received intracranial injection of scrambled vivo-morpholino at P8 (15 mg/kg of body weight of mice). " "The intracranial injection was given manually in 8-day-old BALB/c mice pups (P8) at a single site as vivo-morpholino is believed to diffuse in the tissue [35, 36]."
Cheatham AM, Davis SE, Khatua AK, Popik W. Blocking the 5′ splice site of exon 4 by a morpholino oligomer triggers APOL1 protein isoform switch. Sci Rep. 2018;8:8739 doi:10.1038/s41598-018-27104-x
Delivery to cell culture: AB8/13 podocytes
Mukherjee S, Akbar I, Kumari B, Vrati S, Basu A, Banerjee A. Japanese Encephalitis Virus Infected Microglial Cells Secrete Exosomes Containing let-7a/b that Facilitate Neuronal Damage via Caspase Activation. bioRxiv. 2018. doi:10.1101/324715
12.5 mg/kg delivered by intracranial injection to 10-day-old BALB/c mice pups (TLR-7 knockdown)
Checa-Rojas A, Delgadillo-Silva LF, Velasco-Herrera MDC, Andrade-Domínguez A, Gil J, Santillán O, Lozano L, Toledo-Leyva A, Ramírez-Torres A, Talamas-Rohana P, Encarnación-Guevara S. GSTM3 and GSTP1: novel players driving tumor progression in cervical cancer. Oncotarget. 2018 Apr 24;9(31):21696-21714. doi: 10.18632/oncotarget.24796. eCollection 2018 Apr 24.
cell culture: HeLa, HaCaT, SiHA, CC; murine xenograft: SiHa, CaSki, HeLa, CaLo, MDA, CoLo. The paper does not state that these are Vivo-Morpholinos, but Gene Tools has only manufactured these sequences as Vivo-Morpholinos.
Wyatt EJ, Demonbreun AR, Kim EY, Puckelwartz MJ, Vo AH, Dellefave-Castillo LM, Gao QQ, Vainzof M, Pavanello RCM, Zatz M, McNally EM. Efficient exon skipping of SGCG mutations mediated by phosphorodiamidate morpholino oligomers. JCI Insight. 2018 May 3;3(9). pii: 99357. doi: 10.1172/jci.insight.99357. [Epub ahead of print]
human primary fibroblasts differentiated to myogenic cells
Popik W, Khatua A, Hildreth JEK, Lee B, Alcendor DJ. Phosphorodiamidate morpholino targeting the 5' untranslated region of the ZIKV RNA inhibits virus replication. Virology. 2018 Apr 18;519:77-85. doi: 10.1016/j.virol.2018.04.001. [Epub ahead of print]
cell culture: human glomerular podocytes
Tampe B, Tampe D, Nyamsuren G, Klöpper F, Rapp G, Kauffels A, Lorf T, Zeisberg EM, Müller GA, Kalluri R, Hakroush S, Zeisberg M. Pharmacological induction of hypoxia-inducible transcription factor ARNT attenuates chronic kidney failure. J Clin Invest. 2018 Apr 17. pii: 89632. doi: 10.1172/JCI89632. [Epub ahead of print]
Vivo-Morpholino 12.5 mg/kg i.p. injection in mice every other day starting two days prior of surgery
Zhou D, Shao Q, Fan X, Wu P, Lin W, Wei H, He F, Jiang Y. Regulation of Tak1 alternative splicing by splice-switching oligonucleotides. Biochem Biophys Res Commun. 2018 Feb 20. pii: S0006-291X(18)30397-8. doi: 10.1016/j.bbrc.2018.02.160. [Epub ahead of print]
"The Vivo-morpholino SSOs showed significant splice-switching of Tak1 in mouse liver, with a persistence of at least 10 days after initial SSOs delivery."
Veltrop M, van Vliet L, Hulsker M, Claassens J, Brouwers C, Breukel C, van der Kaa J, Linssen MM, den Dunnen JT, Verbeek S, Aartsma-Rus A, van Putten M. A dystrophic Duchenne mouse model for testing human antisense oligonucleotides. PLoS ONE. 2018;13(2):e0193289. doi:10.1371/journal.pone.0193289
Popek M, Bobula B, Sowa J, Hess G, Albrecht J, Zielinska M. Influence of astrocytic glutamine transporter SN1 deficiency on electrophysiological correlates of glutamatergic transmission. J Neurochem. 2017; 142(suppl 1):125. doi:10.1111/jnc.14093
Kim S, Alsrhani A, Zafreen L, Khandekar G, Marlow FL, Abrams EW, Mullins MC, Jagadeeswaran P. G protein-coupled receptor gpr34l mutation affects thrombocyte function in zebrafish. Br J Haematol. 2017 Dec 21. doi: 10.1111/bjh.15046. [Epub ahead of print]
iv injection into zebrafish
Phumesin P, Junking M, Panya A, Yongpitakwattana P, Noisakran S, Limjindaporn T, Yenchitsomanus P. Vivo-morpholino oligomers strongly inhibit dengue virus replication and production. Arch Virol. 2017;[Epub ahead of print] doi:10.1007/s00705-017-3666-9
cell culture: Vero, A549, U937, primary human monocytes
Adamo P, Porazinski S, Rajatileka S, Jumbe S, Hagen R, Cheung MK, Wilson I, Ladomery MR. The oncogenic transcription factor ERG represses the transcription of the tumour suppressor gene PTEN in prostate cancer cells. Oncol Lett. 2017 Nov;14(5):5605-5610. doi: 10.3892/ol.2017.6841. Epub 2017 Aug 28.
Vivo-Morpholinos combined with Endo-Porter in VCaP (ATCC CRL-2876) cell culture
Lu Y, Hu Z, Mangala LS, Stine ZE, Hu X, Jiang D, Xiang Y, Zhang Y, Pradeep S, Rodriguez-Aguayo C, Lopez-Berestein G, Demarzo A, Sood AK, Zhang L, Dang CV. MYC targeted long non-coding RNA DANCR promotes cancer in part by reducing p21 levels. Cancer Res. 2017;[Epub ahead of print] doi:10.1158/0008-5472.
Targeting lncRNA in P493-6 cell culture
Radhakrishnan U, Alsrhani A, Sundaramoorthi H, Khandekar G, Kashyap M, Fuchs JL, Perkins BD, Omori Y, Jagadeeswaran P. Intraflagellar transport proteins are involved in thrombocyte filopodia formation and secretion. Platelets. 2017 Nov 10:1-10. doi: 10.1080/09537104.2017.1361524. [Epub ahead of print]
zebrafish
Wan Y, McDaniel K, Wu N, Ramos-Lorenzo S, Glaser T, Venter J, Francis H, Kennedy L, Sato K, Zhou T, Kyritsi K, Huang Q, Annable T, Wu C, Glaser S, Alpini G, Meng F. Regulation of Cellular Senescence by miR-34a in Alcoholic Liver Injury. Am J. Pathol. 2017;[Epub ahead of print] doi:10.1016/j.ajpath.2017.08.027
mice with Vivo-Morpholino targeting liver miR-34a
Sindhwani A, Arya SB, Kaur H, Jagga D, Tuli A, Sharma M. Salmonella exploits the host endolysosomal tethering factor HOPS complex to promote its intravacuolar replication. PLoS Pathog. 2017;13(10):e1006700. doi:10.1371/journal.ppat.1006700
mice injected i.v. with 12.5 mg/Kg vivo-morpholino for two consecutive days at an interval of 24 hr
de Morrée A, van Velthoven CTJ, Gan Q, Salvi JS, Klein JDD, Akimenko I, Quarta M, Biressi S, Rando TA. Staufen1 inhibits MyoD translation to actively maintain muscle stem cell quiescence. PNAS USA. 2017;114(43):E8996-E9005 doi:10.1073/pnas.1708725114
mice
Kirschner KM, Sciesielski LK, Krueger K, Scholz H. Wilms tumor protein-dependent transcription of VEGF receptor 2 and hypoxia regulate expression of the testis-promoting gene Sox9 in murine embryonic gonads. J Biol Chem. 2017 Oct 17. pii: jbc.M117.816751. doi: 10.1074/jbc.M117.816751. [Epub ahead of print]
explant culture: murine gonads, kidneys
Touma M, Kang X, Gao F, Zhao Y, Cass AA, Biniwale R, Xiao X, Eghbali M, Coppola G, Reemtsen B, Wang Y. Wnt11 regulates cardiac chamber development and disease during perinatal maturation. JCI Insight. 2017 Sep 7;2(17). pii: 94904. doi: 10.1172/jci.insight.94904. eCollection 2017 Sep 7.
mice: maternal i.v. for embryo delivery of Vivo-Morpholino
Wehner D, Tsarouchas TM, Michael A, Haase C, Weidinger G, Reimer MM, Becker T, Becker CG. Wnt signaling controls pro-regenerative Collagen XII in functional spinal cord regeneration in zebrafish. Nat Comm. 2017;8:126. doi:10.1038/s41467-017-00143-0
Two Vivo-Morpholinos at 0.25 mM each, 9 injections of 3 nl per injection (27 nl total), injected into cardiac ventricle of 3dpf larvae.
Glennon EKK, Torrevillas BK, Morrissey SF, Ejercito JM, Luckhart S. Abscisic acid induces a transient shift in signaling that enhances NF-κB-mediated parasite killing in the midgut of Anopheles stephensi without reducing lifespan or fecundity. Parasit Vectors. 2017 Jul 13;10(1):333. doi: 10.1186/s13071-017-2276-4.
Vivo-Morpholinos fed to mosquitos, delivery to the midgut
Marquardt A, Al-Dabet MM, Ghosh S, Kohli S, Manoharan J, ElWakiel A, Gadi I, Bock F, Nazir S, Wang H, Lindquist JA, Nawroth PP, Madhusudhan T, Mertens PR, Shahzad K, Isermann B. Farnesoid X Receptor Agonism Protects against Diabetic Tubulopathy: Potential Add-On Therapy for Diabetic Nephropathy. J Am Soc Nephrol. 2017 Jul 10. pii: ASN.2016101123. doi: 10.1681/ASN.2016101123. [Epub ahead of print]
mice
Bist P, Cheong WS, Ng A, Dikshit N, Kim BH, Pulloor NK, Khameneh HJ, Hedl M, Shenoy AR, Balamuralidhar V, Malik NBA, Hong M, Neutzner A, Chin KC, Kobayashi KS, Bertoletti A, Mortellaro A, Abraham C, MacMicking JD, Xavier RJ, Sukumaran B. E3 Ubiquitin ligase ZNRF4 negatively regulates NOD2 signalling and induces tolerance to MDP. Nat Commun. 2017 Jun 28;8:15865. doi: 10.1038/ncomms15865.
7-week-old C57BL/6J mice injected iv at 12.5 mg kg^−1 body weight for three consecutive days
Witola WH, Zhang X, Kim CY. Targeted gene knockdown validates the essential role of lactate dehydrogenase in Cryptosporidium parvum. Int J Parasitol. 2017 Jun 9. pii: S0020-7519(17)30166-2. doi: 10.1016/j.ijpara.2017.05.002. [Epub ahead of print]
Hall C, Ehrlich L, Meng F, Invernizzi P, Bernuzzi F, Lairmore TC, Alpini G, Glaser S. Inhibition of microRNA-24 increases liver fibrosis by enhanced menin expression in Mdr2-/- mice. J Surg Res. 2017 May 11. pii: S0022-4804(17)30273-1. doi: 10.1016/j.jss.2017.05.020. [Epub ahead of print]
Kumar V, Soni UK, Maurya VK, Singh K, Jha RK. Integrin beta8 (ITGB8) activates VAV-RAC1 signaling via FAK in the acquisition of endometrial epithelial cell receptivity for blastocyst implantation. Sci Rep. 2017;7:1885. doi:10.1038/s41598-017-01764-7
Lacrosse AL, O'Donovan SM, Sepulveda-Orengo MT, McCullumsmith RE, Reissner KJ, Schwendt M, Knackstedt LA. Contrasting the role of xCT and GLT-1 upregulation in the ability of ceftriaxone to attenuate the cue-induced reinstatement of cocaine-seeking and normalize AMPA receptor subunit expression. J Neurosci. 2017;[Epub ahead of print] doi:10.1523/JNEUROSCI.3717-16.2017
adult male Sprague Dawley rats injected with Vivo-Morpholino in PBS into nucleus accumbens, 30 pmol/side in 1µl though 33 gauge microinjectors fit though cannulae at 0.5µl/min, daily for three days.
Kyritsi K, Meng F, Zhou T, Wu N, Venter J, Francis H, Kennedy L, Onori P, Franchitto A, Bernuzzi F, Invernizzi P, McDaniel K, Mancinelli R, Alvaro D, Gaudio E, Alpini G, Glaser S. Knockdown of Hepatic Gonadotropin-Releasing Hormone by Vivo-Morpholino Decreases Liver Fibrosis in Multidrug Resistance Gene 2 Knockout Mice by Down-Regulation of miR-200b. Am J Pathol. 2017;[Epub ahead of print] doi:10.1016/j.ajpath.2017.03.013
Duque-Wilckens N, Steinman M, Grinevich V, Trainor B. The Role of Oxytocin Neurons in the Bed Nucleus of the Stria Terminalis in Mediating Social Withdrawal. Biol Psychiat. 2017;81(10)suppl:S44-S45
Rudigier LJ, Dame C, Scholz H, Kirschner KM. Ex vivo cultures combined with vivo-morpholino induced gene knockdown provide a system to assess the role of WT1 and GATA4 during gonad differentiation. PLoS ONE. 2017;12(4):e0176296. doi:10.1371/journal.pone.0176296
knockdown in murine gonad explants
Xu J, Lu Z, Narayan A, Le Rouzic VP, Xu M, Hunkele A, Brown TG, Hoefer WF, Rossi GC, Rice RC, Martínez-Rivera A, Rajadhyaksha AM, Cartegni L, Bassoni DL, Pasternak GW, Pan Y-X. Alternatively spliced mu opioid receptor C termini impact the diverse actions of morphine. J Clin Invest. 2017;[Epub ahead of print] doi:10.1172/JCI88760
"CD-1 mice were i.c.v. administered daily for 4 days (days 1–4) with 10 μg of an ANT-vMO"
Smith AM, Maguire-Nguyen KK, Rando TA, Zasloff MA, Strange KB, Yin VP. The protein tyrosine phosphatase 1B inhibitor MSI-1436 stimulates regeneration of heart and multiple other tissues. npj Regen Med. 2017;2:4. doi:10.1038/s41536-017-0008-1
ip injection in zebrafish
Ansseau E, Vanderplanck C, Wauters A, Harper SQ, Coppée F, Belayew A. Antisense Oligonucleotides Used to Target the DUX4 mRNA as Therapeutic Approaches in FaciosScapuloHumeral Muscular Dystrophy (FSHD). Genes. 2017;8(3):93 doi:10.3390/genes8030093
Roy SR, Wang J, Rana MR, Nakashima M, Tokumoto T. Characterization of membrane progestin receptor α (mPRα) of the medaka and role in the induction of oocyte maturation. Biomed Res. 2017;38(1):79-87. doi: 10.2220/biomedres.38.79.
Injection into abdominal cavity for delivery into oocytes
Hazra B, Kumawat KL, Basu A. The host microRNA miR-301a blocks the IRF1-mediated neuronal innate immune response to Japanese encephalitis virus infection. Sci Signal. 2017;10(466):eaaf5185. doi:10.1126/scisignal.aaf5185
BALB/c mice, intracranially treated targeting miR-301a
McMillin M, Frampton G, Grant S, DeMorrow S. The Neuropeptide Galanin Is Up-Regulated during Cholestasis and Contributes to Cholangiocyte Proliferation. Am J Pathol. 2017;[Epub ahead of print] doi:10.1016/j.ajpath.2016.12.015
rat
Mueller AA, van Velthoven CT, Fukumoto KD, Cheung TH, Rando TA. Intronic polyadenylation of PDGFRα in resident stem cells attenuates muscle fibrosis. Nature. 2016 Nov 28. doi: 10.1038/nature20160. [Epub ahead of print]
Vivo-MO in mouse targeting intronic poly-A site, another for a splice site.
Cruse G, Yin Y, Fukuyama T, Desai A, Arthur GK, Bäumer W, Beaven MA, Metcalfe DD. Exon skipping of FcεRIβ eliminates expression of the high-affinity IgE receptor in mast cells with therapeutic potential for allergy. Proc Natl Acad Sci U S A. 2016 Nov 21. pii: 201608520. [Epub ahead of print]
Used with mouse in vivo allergic dermatitis model.
Wang X, Dunlap KA, Satterfield MC, Wu G, Bazer FW. In vivo knockdown of FGFR2 and MET mRNA in trophectoderm of ovine conceptuses retards their development via abrogation of MAPK and MTOR pathways. J Animal Sci. 2016;94(supp 5):376.
Delivery to Ovis aries (sheep) conceptus.
Chang RM, Xiao S, Lei X, Yang H, Fang F, Yang LY. miRNA-487a Promotes Proliferation and Metastasis in Hepatocellular Carcinoma. Clin Cancer Res. 2016 Nov 8. pii: clincanres.0851.2016. [Epub ahead of print]
miRNA-487a targeted in mice.
Meng S, Matrone G, Jie Lv J, Chen K, Wong WK, Cooke JP. LIM Domain Only 2 Regulates Endothelial Proliferation, Angiogenesis, and Tissue Regeneration. J Am Heart Assoc. 2016 Oct 6;5(10). pii: e004117
Vivo‐Morpholino injections in adult zebrafish: "Briefly, zebrafish were anesthetized in tricaine 0.05 mg/mL, and 2 μL of 0.1 mmol/L vivo‐Mo solution was loaded in a glass capillary and injected with a standard microinjector (IM300 Microinjector; Narishige, Tokyo, Japan) on days 0, 2, 4, 6, 8, 10, 12, and 14." "Adult Tg(fli1:egfp)y1 zebrafish fish (2.5 months old) received a retro‐orbital injection of PBS solution containing lmo2 or control (CT) vivo‐Mo..."
Paveliev M, Fenrich KK, Kislin M, Kuja-Panula J, Kulesskiy E, Varjosalo M, Kajander T, Mugantseva E, Ahonen-Bishopp A, Khiroug L, Kulesskaya N, Rougon G, Rauvala H. HB-GAM (pleiotrophin) reverses inhibition of neural regeneration by the CNS extracellular matrix. Sci Rep. 2016;6:33916. doi:10.1038/srep33916
cultured cells: mouse E17 cortical neurons "E17 cortical neurons cultured for 3 days with the control vivo-morpholino in g and i, or with the vivo-morpholino inhibiting splicing of PTPσ in h and i (4 μg/ml each)"
Wang K-C, Yeh Y-T, Nguyen P, Limqueco E, Lopez J, Thorossian S, Guan K-L, Li Y-SJ, Chien S. Flow-dependent YAP/TAZ activities regulate endothelial phenotypes and atherosclerosis. Proc Nat Acad Sci USA. 2016;[Epub ahead of print] doi:10.1073/pnas.1613121113
"To inhibit YAP/TAZ translation in vivo, the MO-YT or MO-Ctrl diluted in saline were administered at a dose of 10 nmol (~5 mg/kg) through tail-vein injections at 72 h before the PL, immediately after the PL, and twice a week until killing."
Presslauer C, Bizuayehu TT, Razmi K, Fernandes JM, Babiak I. See-Thru-Gonad zebrafish line: developmental and functional validation. Reproduction. 2016 Nov;152(5):507-17. doi: 10.1530/REP-16-0328.
Injection into maturing ovaries.
Lutgen V, Narasipura SD, Sharma A, Min S, Al-Harthi L. β-Catenin signaling positively regulates glutamate uptake and metabolism in astrocytes. J Neuroinflammation. 2016 Sep 10;13(1):242.
Brain injection in mice. "Bilateral injections of vivo-morpholinos (Gene Tools, LLC) were targeted to the mPFC with coordinates relative to Bregma +2.0 A/P, +0.3 M/L, −1.0 D/V ... All morpholinos were at a concentration of 500 nM per 1.0 μl. Mice received 1 μl/hemisphere microinjections on day 1 and day 3."
Rouhiainen A, Zhao X, Vanttola P, Qian K, Kulesskiy E, Kuja-Panula J, Gransalke K, Grönholm M, Unni E, Meistrich M, Tian L, Auvinen P, Rauvala H. HMGB4 is expressed by neuronal cells and affects the expression of genes involved in neural differentiation. Sci Rep. 2016;6:32960. doi:10.1038/srep32960
Gujar R, Maurya N, Yadav V, Gupta M, Arora S, Khatri N, Sen P. c-Src Suppresses Dendritic Cell Antitumor Activity via T Cell Ig and Mucin Protein-3 Receptor. J Immunol. 2016 Sep 1;197(5):1650-62. doi: 10.4049/jimmunol.1600104.
"Mice were then injected i.v. with nontargeting control vivo-morpholino ... or c-Src-targeting vivo-morpholino ... at 12.5 mg/kg body weight or with PBS for 5 consecutive d (day 21-25 posttumor inoculation)."
Personius KE, Slusher BS, Udin SB. Neuromuscular NMDA Receptors Modulate Developmental Synapse Elimination. J Neurosci. 2016 Aug 24;36(34):8783-9. doi: 10.1523/JNEUROSCI.1181-16.2016.
Koh DX, Sng JC. HDAC1 negatively regulates Bdnf and Pvalb required for parvalbumin interneuron maturation in an experience-dependent manner. J Neurochem. 2016 Aug 17. doi: 10.1111/jnc.13773. [Epub ahead of print]
mice
Witola WH, Cooks-Fagbodun S, Reyes Ordonez AR, Matthews K, Abugri DA, McHugh M. Knockdown of phosphoethanolamine transmethylation enzymes decreases viability of Haemonchus contortus. Vet Parasitology. 2016;223:1-6. doi:10.1016/j.vetpar.2016.04.008.
Hildyard JC, Wells DJ. Investigating Synthetic Oligonucleotide Targeting of Mir31 in Duchenne Muscular Dystrophy. PLoS Curr. 2016 Jun 16;8. pii: ecurrents.md.99d88e72634387639707601b237467d7. doi: 10.1371/currents.md.99d88e72634387639707601b237467d7.
cell culture, mdx mice
Miskew Nichols B, Aoki Y, Kuraoka M, Lee JJ, Takeda S, Yokota T. Multi-exon Skipping Using Cocktail Antisense Oligonucleotides in the Canine X-linked Muscular Dystrophy. J Vis Exp. 2016 May 24;(111). doi: 10.3791/53776.
Kotb AM, Simon O, Blumenthal A, Vogelgesang S, Dombrowski F, Amann K, Zimmermann U, Endlich K, Endlich N. Knockdown of ApoL1 in Zebrafish Larvae Affects the Glomerular Filtration Barrier and the Expression of Nephrin. PLoS One. 2016 May 3;11(5):e0153768. doi: 10.1371/journal.pone.0153768. eCollection 2016.
"For in vivo KD, a volume of approximately 6 nl injection buffer was injected into the cardinal vein of 3dpf larvae."
Costales MG, Rzuczek SG, Disney MD. Comparison of small molecules and oligonucleotides that target a toxic, non-coding RNA. Bioorg Med Chem Lett. 2016 Apr 11. pii: S0960-894X(16)30386-9. doi: 10.1016/j.bmcl.2016.04.025. [Epub ahead of print]
Wu N, Meng F, Invernizzi P, Bernuzzi F, Venter J, Standeford H, Onori P, Marzioni M, Alvaro D, Franchitto A, Gaudio E, Glaser S, Alpini G. The secretin/secretin receptor axis modulates liver fibrosis through changes in TGF-β1 biliary secretion. Hepatology. 2016 Apr 26. doi: 10.1002/hep.28622. [Epub ahead of print]
Nizzardo M, Simone C, Rizzo F, Ulzi G, Ramirez A, Rizzuti M, Bordoni A, Bucchia M, Gatti S, Bresolin N, Comi GP, Corti S. Morpholino-mediated SOD1 reduction ameliorates an amyotrophic lateral sclerosis disease phenotype. Sci Rep. 2016;6:21301. doi:10.1038/srep21301
Unmodified Morpholinos worked better than Vivo-Morpholinos in this mouse-brain application.
Schulman S, Bendapudi P, Sharda A, Chen V, Bellido-Martin L, Jasuja R, Furie BC, Flaumenhaft R, Furie B. Extracellular Thiol Isomerases and Their Role in Thrombus Formation. Antioxid Redox Signal. 2016 Jan 1;24(1):1-15. doi: 10.1089/ars.2015.6530. Epub 2015 Nov 18.
McMillin M, Frampton G, Quinn M, Ashfaq S, de los Santos III M, Grant S, DeMorrow S. Bile Acid Signaling Is Involved in the Neurological Decline in a Murine Model of Acute Liver Failure. Am J. Pathol. 2015;186(2):312–23. doi:10.1016/j.ajpath.2015.10.005
Vivo-Morpholinos delivered by intracerebroventricular infusion into mice.
Dewaele M, Tabaglio T, Willekens K, Bezzi M, Teo SX, Low DHP, Koh CM, Rambow F, Fiers M, Rogiers A, Radaelli E, Al-Haddawi M, Tan SY, Hermans E, Amant F, Yan H, Lakshmanan M, Koumar RC, Lim ST, Derheimer FA, Campbell RM, Bonday Z, Tergaonkar V, Shackleton M, Blattner C, Marine J-C, Guccione E. Antisense oligonucleotide-mediated MDM4 exon 6 skipping impairs tumor growth. J Clin Invest. 2015;[Epub ahead of print] doi:10.1172/JCI82534
Wong TT, Zohar Y. Production of reproductively sterile fish by a non-transgenic gene silencing technology. Sci Rep. 2015 Oct 29;5:15822. doi: 10.1038/srep15822.
"We discovered that a molecular transporter, Vivo, can effectively carry the Morpholino oligomer (MO) across the chorion, enter the embryo and reach target cells".
Yonezawa T, Chen KH, Ghosh MK, Rivera L, Dill R, Ma L, Villa PA, Kawaminami M, Walker AM. Anti-metastatic outcome of isoform-specific prolactin receptor targeting in breast cancer. Cancer Lett. 2015 Sep 28;366(1):84-92. doi: 10.1016/j.canlet.2015.06.010. Epub 2015 Jun 18.
Delivery into mice with human xenograft tumors using inter-scapular subcutaneous Alzet osmotic pump
Garg A, Wesolowski D, Alonso D, Deitsch KW, Ben Mamoun C, Altman S. Targeting protein translation, RNA splicing, and degradation by morpholino-based conjugates in Plasmodium falciparum. Proc Natl Acad Sci U S A. 2015 Sep 8. pii: 201515864. [Epub ahead of print]
Park SA, Platt J, Lee JW, López-Giráldez F, Herbst RS, Koo JS. E2F8 as a Novel Therapeutic Target for Lung Cancer. J Natl Cancer Inst. 2015 Jun 18;107(9). pii: djv151. doi: 10.1093/jnci/djv151. Print 2015 Sep.
"Then, the mice received subcutaneous injections of cells (2 x 106 cells/flank) and were injected intraperitoneally with mo-control or mo-E2F8 (10mg/kg) (vivo morpholino, Gene Tools, LLC) every other day, five times, once the size of the xenograft (n = 8 per group) reached approximately 5 x 5mm (length x width)."
Bednarek D, González-Rosa JM, Guzmán-Martínez G, Gutiérrez-Gutiérrez Ó, Aguado T, Sánchez-Ferrer C, Marques IJ, Galardi-Castilla M, de Diego I, Gómez MJ, Cortés A, Zapata A, Jiménez-Borreguero LJ, Mercader N, Flores I. Telomerase Is Essential for Zebrafish Heart Regeneration. Cell Rep. 2015 Sep 8;12(10):1691-703. doi: 10.1016/j.celrep.2015.07.064. Epub 2015 Aug 28.
Delivery into zebrafish hearts.
Grande MT, Sánchez-Laorden B, López-Blau C, De Frutos CA, Boutet A, Arévalo M, Rowe RG, Weiss SJ, López-Novoa JM, Nieto MA. Snail1-induced partial epithelial-to-mesenchymal transition drives renal fibrosis in mice and can be targeted to reverse established disease. Nat Med. 2015 Sep;21(9):989-97. doi: 10.1038/nm.3901. Epub 2015 Aug 3.
Salewsky B, Hildebrand G, Rothe S, Parplys AC, Radszewski J, Kieslich M, Wessendorf P, Krenzlin H, Borgmann K, Nussenzweig A, Sperling K, Digweed M. Directed Alternative Splicing in Nijmegen Breakage Syndrome: Proof of Principle Concerning Its Therapeutical Application. Mol Ther. 2015 Aug 12. doi: 10.1038/mt.2015.144. [Epub ahead of print]
Lam S, Chen H, Chen CK, Min N, Chu JJH. Antiviral Phosphorodiamidate Morpholino Oligomers are Protective against Chikungunya Virus Infection on Cell-based and Murine Models. Sci Rep. 2015;5:12727 doi:10.1038/srep12727
Cator LJ, Pietri JE, Murdock CC, Ohm JR, Lewis EE, Read AF, Luckhart S, Thomas MB. Immune response and insulin signalling alter mosquito feeding behaviour to enhance malaria transmission potential. Sci Rep. 2015;5:11947 doi:10.1038/srep11947
Vivo-Morpholinos fed to mosquitos for gut knockdown
Carlson CG, Stein L, Dole E, Potter RM, Bayless D. Agents Which Inhibit NF-κB Signaling Block Spontaneous Contractile Activity and Negatively Influence Survival of Developing Myotubes. J Cell Physiol. 2015 Jun 30. doi: 10.1002/jcp.25085. [Epub ahead of print]
cell culture: murine primary myotubes
Xiang Y, Stine ZE, Xia J, Lu Y, O’Connor RS, Altman BJ, Hsieh AL, Gouw AM, Thomas AG, Gao P, Sun L, Song L, Yan B, Slusher BS, Zhuo J, Ooi LL, Lee CGL, Mancuso A, McCallion AS, Le A, Milone MC, Rayport S, Felsher DW, Dang CV. Targeted inhibition of tumor-specific glutaminase diminishes cell-autonomous tumorigenesis. J Clin Invest. 2015;[Epub ahead of print] doi:10.1172/JCI75836.
"Knockdown of human GLS gene was carried out with the GLS NMD Vivo-Morpholino obtained from Gene Tools. Equal volumes (60 μl) of PBS and standard control (nontargeting) Vivo-Morpholino (Gene Tools) were used as negative controls. Vivo-Morpholino or vehicle (PBS) was injected subcutaneously next to (~0.1 to 0.5 cm from the tumor), but not into, the xenografts every other day for 11 to 13 days."
Ray D, Han Y, Franchitto A, DeMorrow S, Meng F, Venter J, McMillin M, Kennedy L, Francis H, Onori P, Mancinelli R, Gaudio E, Alpini G, Glaser SS. Gonadotropin-releasing hormone stimulates biliary proliferation by paracrine/autocrine mechanisms. Am J Pathol. 2015 Apr;185(4):1061-72. doi: 10.1016/j.ajpath.2014.12.004.
Kann M, Bae E, Lenz MO, Li L, Trannguyen B, Schumacher VA, Taglienti ME, Bordeianou L, Hartwig S, Rinschen MM, Schermer B, Benzing T, Fan CM, Kreidberg JA. WT1 targets Gas1 to maintain nephron progenitor cells by modulating FGF signals. Development. 2015 Apr 1;142(7):1254-66. doi: 10.1242/dev.119735.
Kumar V, Maurya VK, Joshi A, Meeran SM, Jha RK. Integrin Beta 8 (ITGB8) Regulates Embryo Implantation Potentially via Controlling the Activity of TGF-B1 in Mice. Biol Reprod. 2015 Mar 18. pii: biolreprod.114.122838. [Epub ahead of print] Vivo-Morpholino injected into uterine horn
Gramlich M, Pane LS, Zhou Q, Chen Z, Murgia M, Schötterl S, Goedel A, Metzger K, Brade T, Parrotta E, Schaller M, Gerull B, Thierfelder L, Aartsma-Rus A, Labeit S, Atherton JJ, McGaughran J, Harvey RP, Sinnecker D, Mann M, Laugwitz K-L, Gawaz MP, Moretti A. Antisense-mediated exon skipping: a therapeutic strategy for titin-based dilated cardiomyopathy . EMBO Mol Med. 2015;[Epub ahead of print] doi:10.15252/emmm.201505047
Vivo-Morpholinos i.p. in mice
Koganti SR, Zhu Z, Subbotina E, Gao Z, Sierra A, Proenza M, Yang L, Alekseev A, Hodgson-Zingman D, Zingman L. Disruption of KATP channel expression in skeletal muscle by targeted oligonucleotide delivery promotes activity-linked thermogenesis. Mol Ther. 2015 Feb 4. doi: 10.1038/mt.2015.21. [Epub ahead of print]
Vivo-Morpholinos injected into leg muscle in mice
Echigoya Y, Aoki Y, Miskew B, Panesar D, Touznik A, Nagata T, Tanihata J, Nakamura A, Nagaraju K, Yokota T. Long-Term Efficacy of Systemic Multiexon Skipping Targeting Dystrophin Exons 45-55 With a Cocktail of Vivo-Morpholinos in Mdx52 Mice. Mol Ther Nucleic Acids. 2015 Feb 3;4:e225. doi: 10.1038/mtna.2014.76.
Ivanova AS, Shandarin IN, Ermakova GV, Minin AA, Tereshina MB, Zaraisky AG. The secreted factor Ag1 missing in higher vertebrates regulates fins regeneration in Danio rerio. Sci Rep. 2015 Jan 29;5:8123. doi: 10.1038/srep08123.
McClelland KS, Wainwright EN, Bowles J, Koopman P. Rapid screening of gene function by systemic delivery of morpholino oligonucleotides to live mouse embryos. PLoS One. 2015 Jan 28;10(1):e0114932. doi: 10.1371/journal.pone.0114932. eCollection 2015.
Rodrigues-Diez RR, Garcia-Redondo AB, Orejudo M, Rodrigues-Diez R, Briones AM, Bosch-Panadero E, Kery G, Pato J, Ortiz A, Salaices M, Egido J, Ruiz-Ortega M. The C-terminal module IV of connective tissue growth factor, through EGFR/Nox1 signaling, activates the NF-κB pathway and proinflammatory factors in vascular smooth muscle cells. Antioxid Redox Signal. 2015 Jan 1;22(1):29-47. doi: 10.1089/ars.2013.5500.
Cutler CP, Tanner P. Successful Knockdown of Aquaporin 1 (AQP1) Protein Expression in American Eel (Anguilla rostrata) Gastrointestinal Tract Using Direct-Infusions of Vivo-Morpholinos. The Bulletin: Mount Desert Island Biological Laboratory. 2014;53:12-13.
Hegde V, Hickerson RP, Nainamalai S, Campbell PA, Smith FJ, McLean WH, Pedrioli DM. In vivo gene silencing following non-invasive siRNA delivery into the skin using a novel topical formulation. J Control Release. 2014 Dec 28;196:355-62. doi: 10.1016/j.jconrel.2014.10.022. Epub 2014 Oct 30.
mouse intradermal paw injection
Heyland A, Hodin J, Bishop C. Manipulation of Developing Juvenile Structures in Purple Sea Urchins (Strongylocentrotus purpuratus) by Morpholino Injection into Late Stage Larvae. PLoS One. 2014 Dec 1;9(12):e113866. doi: 10.1371/journal.pone.0113866. eCollection 2014.
Stagg BC, Uehara H, Lambert N, Rai R, Gupta I, Radmall B, Bates T, Ambati BK. Morpholino-Mediated Isoform Modulation of Vascular Endothelial Growth Factor Receptor-2 (VEGFR2) Reduces Colon Cancer Xenograft Growth. Cancers. 2014;6(4):2330-42. doi:10.3390/cancers6042330
mouse carrying human tumor xenograft
Prasad AA, McNally GP. Effects of Vivo Morpholino Knockdown of Lateral Hypothalamus Orexin/Hypocretin on Renewal of Alcohol Seeking. PLoS ONE. 2014;9(10):e110385. doi:10.1371/journal.pone.0110385
Microinfusion into rat lateral hypothalamus using a syringe pump
Berberoglu MA, Dong Z, Li G, Zheng J, Martinez LCGT, Peng J, Wagle M, Reichholf B, Petritsch C, Li H, Pleasure SJ, Guo S. Heterogeneously Expressed fezf2 Patterns Gradient Notch Activity in Balancing the Quiescence, Proliferation, and Differentiation of Adult Neural Stem Cells. J Neurosci. 2014;34(42):13911-23. doi: 10.1523/JNEUROSCI.1976-14.2014
zebrafish
Gallego-Villar L, Viecelli HM, Pérez B, Harding CO, Ugarte M, Thöny B, Desviat LR. A sensitive assay system to test antisense oligonucleotides for splice suppression therapy in the mouse liver. Mol Ther Nucleic Acids. 2014 Sep 16;3:e193. doi: 10.1038/mtna.2014.44.
Sundaramoorthi H, Khandekar G, Kim S, Jagadeeswaran P. Knockdown of αIIb by RNA degradation by delivering deoxyoligonucleotides piggybacked with control vivo-morpholinos into zebrafish thrombocytes. Blood Cell Molec Dis. 2014;[Epub ahead of print] doi:10.1016/j.bcmd.2014.07.016
Francis H, McDaniel K, Han Y, Liu X, Kennedy L, Yang F, McCarra J, Zhou T, Glaser S, Venter J, Huang L, Levine P, Lai J-M, Liu C-G, Alpini G, Meng F. Regulation of the extrinsic apoptotic pathway by microRNA-21 in alcoholic liver injury. J Biol Chem. 2014;[Epub ahead of print] doi:10.1074/jbc.M114.602383
Sud R, Geller ET, Schellenberg GD. Antisense-mediated Exon Skipping Decreases Tau Protein Expression: A Potential Therapy For Tauopathies. Mol Ther Nucleic Acids. 2014 Jul 29;3:e180. doi: 10.1038/mtna.2014.30.
mice, i.m. injection
McMillin M, Galindo C, Pae HY, Frampton G, Di Patre PL, Quinn M, Whittington E, DeMorrow S. Gli1 Activation and Protection Against Hepatic Encephalopathy is Suppressed by Circulating Transforming Growth Factor β1 in Mice. J Hepatol. 2014;[Epub ahead of print] doi:10.1016/j.jhep.2014.07.015
mice, intra-cortical infusion
Motamedi FJ, Badro DA, Clarkson M, Rita Lecca M, Bradford ST, Buske FA, Saar K, Hübner N, Brändli AW, Schedl A. WT1 controls antagonistic FGF and BMP-pSMAD pathways in early renal progenitors . Nat Commun. 2014 Jul 17;5:4444. doi: 10.1038/ncomms5444.
organ culture: mouse kidney
Laranjeiro R, Whitmore D. Transcription factors involved in retinogenesis are co-opted by the circadian clock following photoreceptor differentiation. Development. 2014 Jul;141(13):2644-56. doi: 10.1242/dev.104380. Epub 2014 Jun 12. "Intraocular injections were performed in anaesthetised 5 dpf larvae with 0.4 nl/eye 0.5 mM neurod translation-blocking vivo-morpholino ... 24-27 h before dissection of larval eyes."
Yuan Y, Zhang J, Chang Q, Zeng J, Xin F, Wang J, Zhu Q, Wu J, Lu J, Guo W, Yan X, Jiang H, Zhou B, Li Q, Gao X, Yuan H, Yang S, Han D, Mao Z, Chen P, Lin X, Dai P. De novo mutation in ATP6V1B2 impairs lysosome acidification and causes dominant deafness-onychodystrophy syndrome. Cell Res. 2014 Jun 10. doi: 10.1038/cr.2014.77. [Epub ahead of print] No abstract available.
Oligos used were Vivo-Morpholinos (confirmed with author)
Nazmi A, Mukherjee S, Kundu K, Dutta K, Mahadevan A, Shankar SK, Basu A. TLR7 is a key regulator of innate immunity against Japanese Encephalitis Virus infection. Neurobiol Dis. 2014 Jun 5. pii: S0969-9961(14)00154-5. doi: 10.1016/j.nbd.2014.05.036. [Epub ahead of print]
Pietri JE, Cheung KW, Luckhart S. Knockdown of mitogen-activated protein kinase (MAPK) signalling in the midgut of Anopheles stephensi mosquitoes using antisense morpholinos. Insect Mol Biol. 2014 May 28. doi: 10.1111/imb.12103. [Epub ahead of print]
Ferguson DP, Dangott JJ, Timothy Lightfoot JT. Lessons learned from vivo-morpholinos: How to avoid vivo-morpholino toxicity. BioTechniques. 2014;56(5):251-6.
Pfefferli C, Müller F, Jaźwińska A, Wicky C. Specific NuRD components are required for fin regeneration in zebrafish. BMC Biology. 2014;(12):30 doi:10.1186/1741-7007-12-30
Vivo-MO injected into clipped zebrafish fins.
Tan CW, Chan YF, Quah YW, Poh CL. Inhibition of enterovirus 71 infection by antisense octaguanidinium dendrimer-conjugated morpholino oligomers. Antiviral Res. 2014;[Epub ahead of print] doi:10.1016/j.antiviral.2014.04.004
Vivo-MO in cell cultures.
Renzi A, Mancinelli R, Onori P, Franchitto A, Alpini G, Glaser S, Gaudio E. Inhibition of the liver expression of arylalkylamine N-acetyltransferase increases the expression of angiogenic factors in cholangiocytes. Hepatobiliary Surg Nutr. 2014;3(1):4-10. doi:10.3978/j.issn.2304-3881.2014.01.02
Shaked I, Hanna DB, Gleißner C, Marsh B, Plants J, Tracy D, Anastos K, Cohen M, Golub ET, Karim R, Lazar J, Prasad V, Tien PC, Young MA, Landay AL, Kaplan RC, Ley K. Macrophage Inflammatory Markers Are Associated With Subclinical Carotid Artery Disease in Women With Human Immunodeficiency Virus or Hepatitis C Virus Infection. Arterioscler Thromb Vasc Biol. 2014 Mar 20. [Epub ahead of print]
Nizzardo N, Simone C, Salani S, Ruepp M-D, Rizzo F, Ruggieri M, Zanetta C, Brajkovic S, Moulton HM, Müehlemann O, Bresolin N, Comi GP, Corti S. Effect of Combined Systemic and Local Morpholino Treatment on the Spinal Muscular Atrophy Δ7 Mouse Model Phenotype. Clin Therap. 2014. 36(3):340-56. doi:10.1016/j.clinthera.2014.02.004
Carraro G, Shrestha A, Rostkovius J, Contreras A, Chao CM, El Agha E, Mackenzie B, Dilai S, Guidolin D, Taketo MM, Günther A, Kumar ME, Seeger W, De Langhe S, Barreto G, Bellusci S. miR-142-3p balances proliferation and differentiation of mesenchymal cells during lung development. Development. 2014 Feb 19. [Epub ahead of print]
Glaser S, Meng F, Han Y, Onori P, Chow BK, Francis H, Venter J, McDaniel K, Marzioni M, Invernizzi P, Ueno Y, Lai JM, Huang L, Standeford H, Alvaro D, Gaudio E, Franchitto A, Alpini G. Secretin Stimulates Biliary Cell Proliferation by Regulating Expression of MicroRNA 125b and MicroRNA let7a in Mice. Gastroenterology. 2014 Feb 25. pii: S0016-5085(14)00241-8. doi: 10.1053/j.gastro.2014.02.030. [Epub ahead of print]
Two tail-vein injections at 30 mg/kg targeting microRNAs mmu-miR-125b or mmu-miR-let7
Reissner KJ, Gipson CD, Tran PK, Knackstedt LA, Scofield MD, Kalivas PW. Glutamate transporter GLT-1 mediates N-acetylcysteine inhibition of cocaine reinstatement. Addic Biol. 2014;[Epub ahead of print] doi:10.1111/adb.12127
Rat nucleus accumbens
Lee TKW, Cheung VCH, Lu P, Lau EYT, Ma S, Tang KH, Tong M, Lo J, Ng IOL. Blockade of CD47 mediated CTSS-PAR2 signaling provides a therapeutic target for hepatocellular carcinoma. Hepatology. 2014;[Epub ahead of print] doi:10.1002/hep.27070
Human xenograft in mice, intratumoral injection
Ferguson DP, Dangott LJ, Schmitt EE, Vellers HL, Lightfoot JT. Differential Skeletal Muscle Proteome of High and Low Active mice. J Appl Physiol. 1985. 2014 Feb 6. [Epub ahead of print]
Karkampouna S, Kruithof BP, Kloen P, Obdeijn MC, van der Laan AM, Tanke HJ, Kemaladewi DU, Hoogaars WM, 't Hoen PA, Aartsma-Rus A, Clark IM, Ten Dijke P, Goumans MJ, Kruithof-de Julio M. Novel Ex Vivo Culture Method for the Study of Dupuytren's Disease: Effects of TGFβ Type 1 Receptor Modulation by Antisense Oligonucleotides. Mol Ther Nucleic Acids. 2014 Jan 21;3:e142. doi: 10.1038/mtna.2013.69.
Human explant cultures
Reissner KJ, Brown RM, Spencer S, Tran PK, Thomas CA, Kalivas PW. Chronic administration of the methylxanthine propentofylline impairs reinstatement to cocaine by a GLT-1-dependent mechanism. Neuropsychopharmacology. 2014 Jan;39(2):499-506. doi: 10.1038/npp.2013.223. Epub 2013 Aug 29.
Velu CS, Chaubey A, Phelan JD, Horman SR, Wunderlich M, Guzman ML, Jegga AG, Zeleznik-Le NJ, Chen J, Mulloy JC, Cancelas JA, Jordan CT, Aronow BJ, Marcucci G, Bhat B, Gebelein B, Grimes HL. Therapeutic antagonists of microRNAs deplete leukemia-initiating cell activity. J Clin Invest. 2014 Jan 2;124(1):222-36. doi: 10.1172/JCI66005. Epub 2013 Dec 16.
Bollinger RJ, Bujak, JK, Madsen SS, Tipsmark CK. Vivo-Morpholino Induced Knock-Down of Gill Na,K-ATPase Impairs Seawater Acclimation in Rainbow Trout. Society for Integrative and Comparative Biology 2014. (poster) P2.136 rxb031@email.uark.edu
Ramachandran A, McGill MR, Xie Y, Ni HM, Ding WX, Jaeschke H. The receptor interacting protein kinase 3 is a critical early mediator of acetaminophen-induced hepatocyte necrosis in mice. Hepatology. 2013 Dec;58(6):2099-108. doi: 10.1002/hep.26547. Epub 2013 Oct 11.
Khandekar G, Jagadeeswaran P. Role of hepsin in factor VII activation in zebrafish. Blood Cells Mol Dis. 2013 Aug 15. doi:pii: S1079-9796(13)00194-0. 10.1016/j.bcmd.2013.07.014. [Epub ahead of print]
Bitar MS, Abdel-Halim SM, Al-Mulla F. Caveolin-1 upregulation in diabetic fibroblasts and wounded tissues: implication for understanding the underlying mechanisms of non-healing diabetic ulcers. Am J Physiol Endocrinol Metab. 2013 Aug 13. [Epub ahead of print]
Gao Z, Cooper TA. Reexpression of pyruvate kinase M2 in type 1 myofibers correlates with altered glucose metabolism in myotonic dystrophy. PNAS USA. 2013; [Epub ahead of print] doi:10.1073/pnas.1308806110
Voskoboynik A, Newman AM, Corey DM, Sahoo D, Pushkarev D, Neff NF, Passarelli B, Koh W, Ishizuka KJ, Palmeri KJ, Dimov IK, Keasar C, Fan HC, Mantalas GL, Sinha R, Penland L, Quake SR, Weissman IL. Identification of a colonial chordate histocompatibility gene. Science. 2013 Jul 26;341(6144):384-7. doi: 10.1126/science.1238036.
Knockdown in Botryllus schlosseri,a colonial urochordate
Spatazza J, Lee HH, Di Nardo AA, Tibaldi L, Joliot A, Hensch TK, Prochiantz A. Choroid-Plexus-Derived Otx2 Homeoprotein Constrains Adult Cortical Plasticity. Cell Rep. 2013 Jun 11. doi:pii: S2211-1247(13)00231-3. 10.1016/j.celrep.2013.05.014. [Epub ahead of print]
Kizil C, Iltzsche A, Kaslin J, Brand M. Micromanipulation of Gene Expression in the Adult Zebrafish Brain Using Cerebroventricular Microinjection of Morpholino Oligonucleotides. J Vis Exp. 2013;(75):e50415, doi:10.3791/50415
Video protocol for cerebroventricular microinjection in adult zebrafish.
Martin NT, Nakamura K, Davies R, Nahas SA, Brown C, Tunuguntla R, Gatti RA, Hu H. ATM-Dependent MiR-335 Targets CtIP and Modulates the DNA Damage Response. PLoS Genet. 2013;9(5):e1003505. doi:10.1371/journal.pgen.1003505
Lymphoblastoid cell lines
Jacobi CLJ, Rudigier LJ, Scholz H, Kirschner KM. Transcriptional regulation by the Wilms tumor protein, Wt1, suggests a role of the metalloproteinase Adamts16 in murine genitourinary development. J Biol Chem. 2013;[Epub ahead of print] doi:10.1074/jbc.M113.464644
Murine organotypic cultures: kidney and gonads
Ferguson DP, Schmitt EE, Lightfoot JT. Vivo-Morpholinos Induced Transient Knockdown of Physical Activity Related Proteins. PLoS ONE. 2013;8(4):e61472. doi:10.1371/journal.pone.0061472
Mouse; bradykinin enhancing brain delivery
Stec DE, Storm MV, Pruett BE, Gousset MU. Antihypertensive Actions of Moderate Hyperbilirubinemia: Role of Superoxide Inhibition. Am J Hypertens. 2013 Mar 12. [Epub ahead of print] doi: 10.1093/ajh/hpt038
Uehara H, Cho Y, Simonis J, Cahoon J, Archer B, Luo L, Das SK, Singh N, Ambati J, Ambati BK. Dual suppression of hemangiogenesis and lymphangiogenesis by splice-shifting morpholinos targeting vascular endothelial growth factor receptor 2 (KDR). FASEB J, 2013 Jan; 27(1):76–85. doi:10.1096/fj.12-213835 .
Kizil C, Kyritsis N, Dudczig S, Kroehne V, Freudenreich D, Kaslin J, Brand M. Regenerative neurogenesis from neural progenitor cells requires injury-induced expression of Gata3. Dev Cell. 2012. 23(6):1230-7. doi: 10.1016/j.devcel.2012.10.014.
Kyritsis N, Kizil C, Zocher S, Kroehne V, Kaslin J, Freudenreich D, Iltzsche A, Brand M. Acute inflammation initiates the regenerative response in the adult zebrafish brain. Science. 2012 Dec 7;338(6112):1353-6. doi: 10.1126/science.1228773.
Lin JF, Pan HC, Ma LP, Shen YQ, Schachner M. The Cell Neural Adhesion Molecule Contactin-2 (TAG-1) Is Beneficial for Functional Recovery after Spinal Cord Injury in Adult Zebrafish. PLoS ONE. 2012;7(12):e52376. doi:10.1371/journal.pone.0052376 Vivo-Morpholino in Gelfoam at spinal cord injury: "Contactin-2 anti-sense MO significantly reduced contactin-2 protein levels by approximately 82% compared with control MO 10 days after MO treatment being set to 100% ... . Even 3 weeks after MO treatment, contactin-2 protein levels continued to remain decreased to a level of 49% (data not shown). It is noteworthy in this context that MO inhibition has been observed to last for more than 6 weeks."
Hughes CE, Radhakrishnan UP, Lordkipanidzé M, Egginton S, Dijkstra JM, Jagadeeswaran P, Watson SP. G6f-Like Is an ITAM-Containing Collagen Receptor in Thrombocytes. PLoS ONE. 2012;7(12):e52622. doi:10.1371/journal.pone.0052622
Vivo-Morpholinos injected into both adult zebrafish (>3 month) and zebrafish embryos (3dpf).
Malerba A, Kang JK, McClorey G, Saleh AF, Popplewell L, Gait MJ, Wood MJA, Dickson G. Dual Myostatin and Dystrophin Exon Skipping by Morpholino Nucleic Acid Oligomers Conjugated to a Cell-penetrating Peptide Is a Promising Therapeutic Strategy for the Treatment of Duchenne Muscular Dystrophy. Mol Ther Nucleic Acids. 2012;1:e62. doi:/10.1038/mtna.2012.54
Frampton G, Ueno Y, Quinn M, McMillin M, Pae HY, Galindo C, Leyva-Illades D, Demorrow S. The novel growth factor, progranulin, stimulates mouse cholangiocyte proliferation via Sirtuin1-mediated inactivation of FOXO1. Am J Physiol Gastrointest Liver Physiol. 2012 Oct 18. [Epub ahead of print]
Vivo-Morpholino injected into mice.
Renzi A, Demorrow S, Onori P, Carpino G, Mancinelli R, Meng F, Venter J, White M, Franchitto A, Francis H, Han Y, Ueno Y, Dusio G, Jensen KJ, Greene JJ, Glaser S, Gaudio E, Alpini G. Modulation of the biliary expression of arylalkylamine N-acetyltransferase alters the autocrine proliferative responses of cholangiocytes. Hepatology. 2012 Oct 18. doi: 10.1002/hep.26105. [Epub ahead of print]
Vivo-Morpholino injected into rats.
Csányi G, Yao M, Rodriguez AI, Al Ghouleh I, Sharifi-Sanjani M, Frazziano G, Xiaojun H, Kelley EE, Isenberg JS, Pagano PJ. Thrombospondin-1 Regulates Blood Flow via CD47 Receptor–Mediated Activation of NADPH Oxidase 1. Arterioscler Thromb Vasc Biol. 2012;[Epub ahead of print] doi:10.1161/ATVBAHA.112.300031
Luo YJ, Su YH. Opposing Nodal and BMP Signals Regulate Left–Right Asymmetry in the Sea Urchin Larva. PLoS Biol. 2012;10(10): e1001402. doi:10.1371/journal.pbio.1001402
Chen SX, Cherry A, Tari PK, Podgorski K, Kwong YKK, Haas K. The Transcription Factor MEF2 Directs Developmental Visually Driven Functional and Structural Metaplasticity. Cell. 2012;151(1):41–55 doi:10.1016/j.cell.2012.08.028
Domínguez-Bendala J, Álvarez-Cubela S, Nieto M, Vargas N, Espino-Grosso P, Sacher VY, Pileggi A, García E, Ricordi C, Inverardi L, Pastori RL. Intracardial Embryonic Delivery of Developmental Modifiers In Utero. Cold Spring Harb Protoc. 2012;doi:10.1101/pdb.prot069427
Vivo-Morpholino targeting miRNA in mouse embryos.
Xu H, Oliveira-Sales EB, McBride F, Liu B, Hewinson J, Toward M, Hendy EB, Graham D, Dominiczak AF, Giannotta M, Waki H, Ascione R, Paton JF, Kasparov S. Upregulation of junctional adhesion molecule-A is a putative prognostic marker of hypertension. Cardiovasc Res. 2012 Aug 22. [Epub ahead of print]
Tail vein injection in juvenile rats.
Chen Y, Londraville R, Brickner S, El-Shaar L, Fankhauser K, Dearth C, Fulton L, Sochacka A, Bhattarai S, Marrs JA, Liu Q. Protocadherin-17 function in zebrafish retinal development. Dev Neurobiol. 2012 Aug 28. doi: 10.1002/dneu.22053. [Epub ahead of print]
Injection into eyes of zebrafish embryos.
Pal D, Dasgupta S, Kundu R, Maitra S, Das G, Mukhopadhyay S, Ray S, Majumdar SS, Bhattacharya S. Fetuin-A acts as an endogenous ligand of TLR4 to promote lipid-induced insulin resistance. Nat Med. 2012 Aug;18(8):1279-85. doi: 10.1038/nm.2851. Epub 2012 Jul 29
Vivo-MO to FetA or Tlr4, 5- to 6-week-old female BALB/c mice, "Twenty-five nmoles of VMO was delivered to tail vein through injection in each mouse for 5 consecutive days, one on each day."
Yokota T, Nakamura A, Nagata T, Saito T, Kobayashi M, Aoki Y, Echigoya Y, Partridge T, Hoffman EP, Takeda S. Extensive and Prolonged Restoration of Dystrophin Expression with Vivo-Morpholino-Mediated Multiple Exon Skipping in Dystrophic Dogs. Nucleic Acid Therapeutics. 2012. [Epub ahead of print] doi:10.1089/nat.2012.0368
Lai BS, Witola WH, El Bissati KE, Zhou Y, Mui E, Fomovska A, McLeod R. Molecular target validation, antimicrobial delivery, and potential treatment of Toxoplasma gondii infections. PNAS. 2012;[Epub ahead of print] doi:10.1073/pnas.1208775109
Terabayashi T, Sakaguchi M, Shinmyozu K, Ohshima T, Johjima A, Ogura T, Miki H, Nishinakamura R. Phosphorylation of Kif26b Promotes Its Polyubiquitination and Subsequent Proteasomal Degradation during Kidney Development. PLoS ONE. 2012;7(6): e39714. doi:10.1371/journal.pone.0039714
"We performed knockdown analysis of Nedd4 in kidney explants from E12.5 embryos...resulted in a slight accumulation of Kif26b after 24 h compared with control-MO, but this increase was not significant and we were also unable to rule out the possibility of off-target effects of the MO (data not shown)."
Aoki Y, Yokota T, Nagata T, Nakamura A, Tanihata J, Saito T, Duguez SMR, Nagaraju K, Hoffman EP, Partridge T, Takeda S. Bodywide skipping of exons 45–55 in dystrophic mdx52 mice by systemic antisense delivery. PNAS. 2012;[Epub ahead of print] doi:10.1073/pnas.1204638109
Cutler CP, Smith D. Vivo-morpholino knockdown of aquaporin 1 (AQP1) protein expression in American eel (Anguilla rostrata) gastrointestinal tract. The Bulletin, MDI Biological Laboratory. 2012;51
Notch EG, Chapline C, Flynn E, Lameyer T, Lowell A, Sato D, Shaw JR, Stanton BA. Mitogen activated protein kinase 14-1 regulates serum glucocorticoid kinase 1 during seawater acclimation in Atlantic killifish, Fundulus heteroclitus. Comp Biochem Physiol A Mol Integr Physiol. 2012 May 2. [Epub ahead of print]
Nazmi A, Mukhopadhyay R, Dutta K, Basu A. STING Mediates Neuronal Innate Immune Response Following Japanese Encephalitis Virus Infection. Sci Rep. 2012;2:347. Epub 2012 Apr 2.
3–4 weeks old BALB/c mice, 5 mg/kg Vivo-Morpholino once per day for five days.
Parra MK, Gallagher TL, Amacher SL, Mohandas N, Conboy JG. Deep intron elements mediate nested splicing events at consecutive AG-dinucleotides to regulate alternative 3' splice site choice in vertebrate 4.1 genes. Mol Cell Biol. 2012 Apr 2. [Epub ahead of print]
Vivo-Morpholinos injected in mouse tail veins at 15mg/kg on two consecutive days.
Sartor GC, Aston-Jones GS. A Septal-Hypothalamic Pathway Drives Orexin Neurons, Which Is Necessary for Conditioned Cocaine Preference. J Neurosci. 2012;32(13):4623-31. doi:10.1523/JNEUROSCI.4561-11.2012
Vivo-Morpholinos administered though cannulae into rat brains.
Owen LA, Uehara H, Cahoon J, Huang W, Simonis J, Ambati BK. Morpholino-Mediated Increase in Soluble Flt-1 Expression Results in Decreased Ocular and Tumor Neovascularization. PLoS ONE. 2012;7(3):e33576. doi:10.1371/journal.pone.0033576
Liu Y, Yu H, Deaton SK, Szaro BG. Heterogeneous Nuclear Ribonucleoprotein K, an RNA-Binding Protein, Is Required for Optic Axon Regeneration in Xenopus laevis. J Neurosci. 2012;32(10):3563-3574. doi:10.1523/JNEUROSCI.5197-11.2012
"In uninjured eyes, it efficiently knocked down hnRNP K expression in only the RGCs, without inducing either an axotomy response or axon degeneration."
Jakobson M, Lintulahti A, Arumäe U. mRNA for N-Bak, a neuron-specific BH3-only splice isoform of Bak, escapes nonsense-mediated decay and is translationally repressed in the neurons. Cell Death Dis. 2012 Feb 2;3:e269. doi: 10.1038/cddis.2012.4.
Cultured mouse neurons
Nakamura K, Du L, Tunuguntla R, Fike F, Cavalieri S, Morio T, Mizutani S, Brusco A, Gatti RA. Functional characterization and targeted correction of ATM mutations identified in Japanese patients with ataxia-telangiectasia. Hum Mutat. 2012 Jan;33(1):198-208. doi: 10.1002/humu.21632. Epub 2011 Nov 9.
Cultured primary cells
Notch EG, Shaw JR, Coutermarsh BA, Dzioba M, Stanton BA. Morpholino Gene Knockdown in Adult Fundulus heteroclitus: Role of SGK1 in Seawater Acclimation. PLoS ONE. 2011;6(12): e29462. doi:10.1371/journal.pone.0029462
Intraperitoneal (IP) injection, 14 µg/g SGK1 translation-blocking vivo-morpholino.
Taniguchi-Ikeda M, Kobayashi K, Kanagawa M, Yu CC, Mori K, Oda T, Kuga A, Kurahashi H, Akman HO, DiMauro S, Kaji R, Yokota T, Takeda S, Toda T. Pathogenic exon-trapping by SVA retrotransposon and rescue in Fukuyama muscular dystrophy. Nature. 2011 Oct 5;478(7367):127-31. doi: 10.1038/nature10456.
Tail-vein and local injections in a mouse model of Fukuyama muscular dystrophy
Kizil C, Brand M. Cerebroventricular Microinjection (CVMI) into Adult Zebrafish Brain Is an Efficient Misexpression Method for Forebrain Ventricular Cells. PLoS ONE. 2011;6(11):e27395. doi:10.1371/journal.pone.0027395
Injection of up to 500 nl into cerebroventricular fluid at Vivo-Morpholino concentrations up to 500 µM.
Osorio FG, Navarro CL, Cadiñanos J, López-Mejía IC, Quirós PM, Bartoli C, Rivera J, Tazi J, Guzmán G, Varela I, Depetris D, de Carlos F, Cobo J, Andrés V, Sandre-Giovannoli AD, Freije JM, Lévy N, López-Otín C. Splicing-Directed Therapy in a New Mouse Model of Human Accelerated Aging. Sci. Transl. Med. 2011;3:106ra107 doi:10.1126/scitranslmed.3002847
6 mg/Kg each of 2 sequences (or 12 mg/Kg control sequence) were administered to progeria model mice by tail vein injection twice per week for 12 weeks, starting at six weeks of age.
Reissner KJ, Sartor GC, Vazey EM, Dunn TE, Aston-Jones G, Kalivas PW. Use of vivo-morpholinos for control of protein expression in the adult rat brain. J Neurosci Meth. 2012 Jan 30;203(2):354-60. doi: 10.1016/j.jneumeth.2011.10.009. Epub 2011 Oct 17.
Intracranial injections in rat brains of oligos targeting three different RNA targets, some though cannula and some by direct injection.
NOTE: Kate Reissner informs us that the doses reported in this paper in nmol should have been reported in pmol; that is, the reported doses are 1000x too high.
Shi YB, Hasebe T, Fu L, Fujimoto K, Ishizuya-Oka A. The development of the adult intestinal stem cells: Insights from studies on thyroid hormone-dependent amphibian metamorphosis. Cell Biosci. 2011 Sep 6;1(1):30.
Injection into stage 53/54 Xenopus tadpoles, knockdown in gut.
Zammarchi F, de Stanchina E, Bournazou E, Supakorndej T, Martires K, Riedel E, Corben AD, Bromberg JF, Cartegni L. Antitumorigenic potential of STAT3 alternative splicing modulation. Proc Natl Acad Sci U S A. 2011 Oct 17. [Epub ahead of print]
Injections of Vivo-Morpholinos in a mouse tumor model either intratumorally at a final concentration of 0.12 mg/30 μl or intravenously at 15 mg/kg. Oligos were administered 2x week for either 1 or 3 weeks.
Nakamura K, Du L, Tunuguntla R, Fike F, Cavalieri S, Morio T, Mizutani S, Brusco A, Gatti RA. Functional characterization and targeted correction of ATM mutations identified in Japanese patients with ataxia-telangiectasia. Hum. Mutat. 2011;[Epub ahead of print] doi:10.1002/humu.21632
Splice correction in lymphoblastoid cell line from patient.
Quinn M, Ueno Y, Pae HY, Huang L, Frampton G, Galindo C, Francis H, Horvat D, McMillin M, Demorrow S. Suppression of the HPA axis during extrahepatic biliary obstruction induces cholangiocyte proliferation in the rat. Am J Physiol Gastrointest Liver Physiol. 2012 Jan 1;302(1):G182-93. doi: 10.1152/ajpgi.00205.2011. Epub 2011 Oct 6.
Intracerebroventricular injection of Vivo-Morpholinos in rat, daily 4ug/rat/day for 7 days.
Taniguchi-Ikeda M, Kobayashi K, Kanagawa M, Yu CC, Mori K, Oda T, Kuga A, Kurahashi H, Akman HO, DiMauro S, Kaji R, Yokota T, Takeda S, Toda T. Pathogenic exon-trapping by SVA retrotransposon and rescue in Fukuyama muscular dystrophy. Nature. 2011;478:127-31 doi:10.1038/nature10456
FCMD model mice, human FCMD lymphoblasts and myotubes.
Vorlová S, Rocco G, LeFave CV, Jodelka FM, Hess K, Hastings ML, Henke H, Cartegni L. Induction of Antagonistic Soluble Decoy Receptor Tyrosine Kinases by Intronic PolyA Activation. Mol Cell. 2011;(43)6:927-39.
Cultured cells: HeLa, MDA-231 & HUVEC.
Nazmi A, Dutta K, Basu A. RIG-I Mediates Innate Immune Response in Mouse Neurons Following Japanese Encephalitis Virus Infection. PLoS ONE. 2011;6(6): e21761. doi:10.1371/journal.pone.0021761
Wu B, Benrashid E, Lu P, Cloer C, Zillmer A, Shaban M, Lu QL. Targeted Skipping of Human Dystrophin Exons in Transgenic Mouse Model Systemically for Antisense Drug Development. PLoS ONE. 2011;6(5):e19906. doi:10.1371/journal.pone.0019906
Kowalik L, Hudspeth AJ. A search for factors specifying tonotopy implicates DNER in hair-cell development in the chick's cochlea. Dev Biol. 2011 Jun 15;354(2):221-31. Epub 2011 Apr 8.
Vivo-Morpholinos locally injected in embryonic chick ears at E6.
Tsonis PA, Haynes T, Maki N, Nakamura K, Casco-Robles MM, Yamada S, Miura T, Chiba C, Del Rio-Tsonis K. Controlling gene loss of function in newts with emphasis on lens regeneration. Nature Protocols. 2011;6:593–599 doi:10.1038/nprot.2011.341 pdf
Widrick JJ, Jiang S, Choi SJ, Knuth ST, Morcos PA. An octaguanidine-morpholino oligo conjugate improves muscle function of mdx mice. Muscle & Nerve 2011 [Epub ahead of print]
Parra MK, Gee S, Mohandas N, Conboy JG. Efficient in vivo manipulation of alternative pre-mRNA splicing events using antisense morpholinos in mice. J Biol Chem. 2011 Feb 25;286(8):6033-9. Epub 2010 Dec 14.
Vivo-Morpholinos evaluated in mice at Lawrence Berkeley National Laboratory.
Kang JK, Malerba A, Popplewell L, Foster K, Dickson G. Antisense-induced Myostatin Exon Skipping Leads to Muscle Hypertrophy in Mice Following Octa guanidine Morpholino Oligomer Treatment. Mol Ther. 2011 Jan;19(1):159-164. doi: 10.1038/mt.2010.212.
Nazmi A, Dutta K, Basu A. Antiviral and Neuroprotective Role of Octaguanidinium Dendrimer-Conjugated Morpholino Oligomers in Japanese Encephalitis. PLoS Negl Trop Dis 2010;4(11): e892. doi:10.1371/journal.pntd.0000892
Viral knockdowns in mice.
Carrillo M, Kim S, Rajpurohit SK, Kulkarni V, Jagadeeswaran P. Zebrafish von Willebrand factor. Blood Cells Mol Dis. 2010 Oct 28. [Epub ahead of print]
Vivo-Morpholinos in adult zebrafish.
Guo Y, Ma L, Cristofanilli M, Hart RP, Hao A, Schachner M. Transcription factor Sox11b is involved in spinal cord regeneration in adult zebrafish. Neuroscience. 2010 Oct 14. [Epub ahead of print]
Vivo-Morpholino on Gelfoam applied to zebrafish spinal cord transection at time of injury.
Matsuda H, Shi YB. An Essential and Evolutionarily Conserved Role of Protein Arginine Methyltransferase 1 for Adult Intestinal Stem Cells During Postembryonic Development. Stem Cells. 2010 Sep 16. [Epub ahead of print]
Intraperitoneal injection in Xenopus laevis.
Azoitei N, Pusapati GV, Kleger A, Möller P, Küfer R, Genze F, Wagner M, van Lint J, Carmeliet P, Adler G, Seufferlein T. Protein kinase D2 is a crucial regulator of tumour cell-endothelial cell communication in gastrointestinal tumours. Gut. 2010 Aug 23. [Epub ahead of print]
Vivo-Morpholinos in mice.
Vera T, Stec DE. Moderate Hyperbilirubinemia Improves Renal Hemodynamics in Angiotensin II Dependent Hypertension. Am J Physiol Regul Integr Comp Physiol. 2010 Oct;299(4):R1044-9. Epub 2010 Jul 28.
Vivo-Morpholinos in mice.
Kreidberg JA. WT1 and kidney progenitor cells. Organogenesis. 2010 Apr;6(2):61-70. Knockdown of WT1 in murine kidney explants; review offering perspective on experiments in Hartwig et al., Development 2010 (below).
Lecce L, Day M, Murphy CR. Abstract 15. ICAM-1 Is Involved in Uterine Receptivity in Rats. The Society for the Study of Reproduction 2010 Annual Meeting.
Maki N, Suetsugu-Maki R, Sano S, Nakamura K, Nishimura O, Tarui H, Del Rio-Tsonis K, Ohsumi K, Agata K, Tsonis PA. Oocyte-type linker histone B4 is required for transdifferentiation of somatic cells in vivo. FASEB J. 2010 Sep;24(9):3462-7. Epub 2010 May 11.
This describes Vivo-Morpholino use in a newt.
Presentations using Vivo-Morpholinos from 2010 American Society of Gene & Cell Therapy meeting:
See abstract 14 on this linked page.
See abstract 16 on this linked page.
Hartwig S, Ho J, Pandey P, Macisaac K, Taglienti M, Xiang M, Alterovitz G, Ramoni M, Fraenkel E, Kreidberg JA. Genomic characterization of Wilms' tumor suppressor 1 targets in nephron progenitor cells during kidney development. Development. 2010 Apr;137(7):1189-203.
Knockdown of WT1 in murine kidney explants.
Chablais F, Jazwinska A. IGF signaling between blastema and wound epidermis is required for fin regeneration. Development. 2010 Mar;137(6):871-9.
Injection of 1.5 mM Vivo-Morpholino into regenerating zebrafish fins.
Kim S, Radhakrishnan UP, Rajpurohit SK, Kulkarni V, Jagadeeswaran P. Vivo-Morpholino knockdown of alphaIIb: A novel approach to inhibit thrombocyte function in adult zebrafish. Blood Cells Mol Dis. 2010 Mar 15;44(3):169-74. Epub 2010 Jan 4.
Knockdown of alphaIIb in thrombocytes by i.v. injection of Vivo-Morpholinos into adult zebrafish.
Pérez B, Rincón A, Jorge-Finnigan A, Richard E, Merinero B, Ugarte M, Desviat LR. Pseudoexon exclusion by antisense therapy in methylmalonic aciduria (MMAuria). Hum Mutat. 2009 Dec;30(12):1676-82.
Report of Vivo-Morpholinos compared with unmodified Morpholinos and Endo-Porter when tested in cultured human fibroblasts.
Moulton JD, Jiang S. Gene Knockdowns in Adult Animals: PPMOs and Vivo-Morpholinos. Molecules. 2009; 14(3):1304-1323.
Wu B, Li YF, Morcos PA, Doran TJ, Lu PJ and Lu QL. Octa-guanidine Morpholino Restores Dystrophin Expression in Cardiac and Skeletal Muscles and Ameliorates Pathology in Dystrophic mdx Mice. Mol. Ther. 2009 May;17(5):864-71. Epub 2009 Mar 10.
Paul A. Morcos, Yongfu Li, and Shan Jiang. Vivo-Morpholinos: A non-peptide transporter delivers Morpholinos into a wide array of mouse tissues. BioTechniques. 2008 Dec;45(6):616-26.
Li YF, Morcos PA. Design and Synthesis of Dendritic Molecular Transporter that Achieves Efficient in Vivo Delivery of Morpholino Antisense Oligo. Bioconjug Chem. 2008 Jul;19(7):1464-70. Epub 2008 Jun 20.
